Clearance of infection with Mycobacterium bovis BCG in mice is enhanced by treatment with S28463 (R-848), and its efficiency depends on expression of wild-type Nramp1 (resistance allele)
- PMID: 11600356
- PMCID: PMC90782
- DOI: 10.1128/AAC.45.11.3059-3064.2001
Clearance of infection with Mycobacterium bovis BCG in mice is enhanced by treatment with S28463 (R-848), and its efficiency depends on expression of wild-type Nramp1 (resistance allele)
Abstract
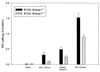
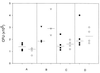
The mouse bcg host resistance gene is known to control the activation of host macrophages for killing of intracellular parasites like Leishmania donovani as well as intracellular bacteria, including Mycobacterium bovis BCG and Salmonella enterica serovar Typhimurium. The Nramp1 gene has been mapped to this locus and affects the efficiency of macrophage activation. It has been shown that imidazoquinoline compounds, including S28463, are able to improve the clearance of a number of intracellular pathogens such as herpes simplex virus 2, human papillomavirus, and Leishmania. The goal of this study was to determine whether S28463 is efficient against infection with another intracellular pathogen, M. bovis BCG, and to determine the molecular basis underlying this effect. To achieve this, B10A.Nramp1(r) and B10A.Nramp1(-/-) mice were infected with M. bovis BCG and treated with S28463. The bacterial content in the spleen from these mice was assayed by a colony-forming assay. In addition, in vitro experiments were performed using bone marrow-derived macrophage cell lines from these mice. These cells were treated with S28463 and/or gamma interferon (IFN-gamma), and nitric oxide (NO) production was measured. Our study was able to show that S28463 acts in synergy with IFN-gamma to increase the production of NO in vitro. We were also able to demonstrate that mice that carried the resistant allele of the Nramp1 gene and were infected with M. bovis BCG responded to treatment with S28463, resulting in a decreased bacterial load after 2 weeks of treatment. Mice that do not express the Nramp1 gene responded only to a very large dose of S28463, and the response was not as efficient as that observed in mice carrying a wild-type Nramp1 allele. Our data provide evidence for the potential of S28463 as an immunomodulator that may be helpful in designing efficient strategies to improve host defense against mycobacterial infection.
Figures


Similar articles
-
Attenuation of MHC class II expression in macrophages infected with Mycobacterium bovis bacillus Calmette-Guérin involves class II transactivator and depends on the Nramp1 gene.J Immunol. 1999 Sep 1;163(5):2688-96. J Immunol. 1999. PMID: 10453010
-
Proteome study of Francisella tularensis live vaccine strain-containing phagosome in Bcg/Nramp1 congenic macrophages: resistant allele contributes to permissive environment and susceptibility to infection.Proteomics. 2002 Jan;2(1):85-93. Proteomics. 2002. PMID: 11788995
-
[Frontier of mycobacterium research--host vs. mycobacterium].Kekkaku. 2005 Sep;80(9):613-29. Kekkaku. 2005. PMID: 16245793 Japanese.
-
Nonspecific resistance to Mycobacteria: the role of the Nramp1 gene.Behring Inst Mitt. 1997 Mar;(99):51-7. Behring Inst Mitt. 1997. PMID: 9303202 Review.
-
Natural resistance to infection with intracellular parasites: molecular genetics identifies Nramp1 as the Bcg/Ity/Lsh locus.J Leukoc Biol. 1995 Oct;58(4):382-90. doi: 10.1002/jlb.58.4.382. J Leukoc Biol. 1995. PMID: 7561513 Review.
References
-
- Ahonen C L, Gibson S J, Smith R M, Pederson L K, Lindh J M, Tomai M A, Vasilakos J P. Dendritic cell maturation and subsequent enhanced T-cell stimulation induced with the novel synthetic immune response modifier R-848. Cell Immunol. 1999;197:62–72. - PubMed
-
- Arany I, Tyring S K, Brysk M M, Stanley M A, Tomai M A, Miller R L, Smith M H, McDermott D J, Slade H B. Correlation between pretreatment levels of interferon response genes and clinical responses to an immune response modifier (imiquimod) in genital warts. Antimicrob Agents Chemother. 2000;44:1869–1873. - PMC - PubMed
-
- Arany I, Tyring S K, Stanley M A, Tomai M A, Miller R L, Smith M H, McDermott D J, Slade H B. Enhancement of the innate and cellular immune response in patients with genital warts treated with topical imiquimod cream 5% Antiviral Res. 1999;43:55–63. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

