Activation of protein kinase C zeta induces serine phosphorylation of VAMP2 in the GLUT4 compartment and increases glucose transport in skeletal muscle
- PMID: 11604519
- PMCID: PMC99955
- DOI: 10.1128/MCB.21.22.7852-7861.2001
Activation of protein kinase C zeta induces serine phosphorylation of VAMP2 in the GLUT4 compartment and increases glucose transport in skeletal muscle
Abstract
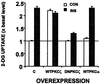
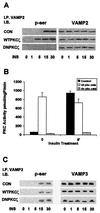
Insulin stimulates glucose uptake into skeletal muscle tissue mainly through the translocation of glucose transporter 4 (GLUT4) to the plasma membrane. The precise mechanism involved in this process is presently unknown. In the cascade of events leading to insulin-induced glucose transport, insulin activates specific protein kinase C (PKC) isoforms. In this study we investigated the roles of PKC zeta in insulin-stimulated glucose uptake and GLUT4 translocation in primary cultures of rat skeletal muscle. We found that insulin initially caused PKC zeta to associate specifically with the GLUT4 compartments and that PKC zeta together with the GLUT4 compartments were then translocated to the plasma membrane as a complex. PKC zeta and GLUT4 recycled independently of one another. To further establish the importance of PKC zeta in glucose transport, we used adenovirus constructs containing wild-type or kinase-inactive, dominant-negative PKC zeta (DNPKC zeta) cDNA to overexpress this isoform in skeletal muscle myotube cultures. We found that overexpression of PKC zeta was associated with a marked increase in the activity of this isoform. The overexpressed, active PKC zeta coprecipitated with the GLUT4 compartments. Moreover, overexpression of PKC zeta caused GLUT4 translocation to the plasma membrane and increased glucose uptake in the absence of insulin. Finally, either insulin or overexpression of PKC zeta induced serine phosphorylation of the GLUT4-compartment-associated vesicle-associated membrane protein 2. Furthermore, DNPKC zeta disrupted the GLUT4 compartment integrity and abrogated insulin-induced GLUT4 translocation and glucose uptake. These results demonstrate that PKC zeta regulates insulin-stimulated GLUT4 translocation and glucose transport through the unique colocalization of this isoform with the GLUT4 compartments.
Figures





References
-
- Bandyopadhyay G, Kanoh Y, Sajan M P, Standaert M L, Farese R V. Effects of adenoviral gene transfer of wild-type, constitutively active, and kinase-defective protein kinase C-lambda on insulin-stimulated glucose transport in L6 myotubes. Endocrinology. 2000;141:4120–4127. - PubMed
-
- Bandyopadhyay G, Standaert M L, Galloway L, Moscat J, Farese R V. Evidence for involvement of protein kinase C (PKC)-zeta and noninvolvement of diacylglycerol-sensitive PKCs in insulin-stimulated glucose transport in L6 myotubes. Endocrinology. 1997;138:4721–4731. - PubMed
-
- Bandyopadhyay G, Standaert M L, Kikkawa U, Ono Y, Moscat J, Farese R V. Effects of transiently expressed atypical (zeta, lambda), conventional (alpha, beta) and novel (delta, epsilon) protein kinase C isoforms on insulin-stimulated translocation of epitope-tagged GLUT4 glucose transporters in rat adipocytes: specific interchangeable effects of protein kinases C-zeta and C-lambda. Biochem J. 1999;337:461–470. - PMC - PubMed
-
- Braiman L, Alt A, Kuroki T, Ohba M, Bak A, Tennenbaum T, Sampson S R. Protein kinase Cdelta mediates insulin-induced glucose transport in primary cultures of rat skeletal muscle. Mol Endocrinol. 1999;13:2002–2012. - PubMed
-
- Braiman L, Alt A, Kuroki T, Ohba M, Bak A, Tennenbaum T, Sampson S R. Insulin induces specific interaction between insulin receptor and PKC in primary cultured skeletal muscle. Mol Endocrinol. 2001;15:565–574. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
