Glycosylation and the activation of proteinase-activated receptor 2 (PAR(2)) by human mast cell tryptase
- PMID: 11606310
- PMCID: PMC1572998
- DOI: 10.1038/sj.bjp.0704303
Glycosylation and the activation of proteinase-activated receptor 2 (PAR(2)) by human mast cell tryptase
Abstract
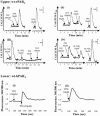
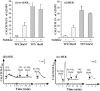
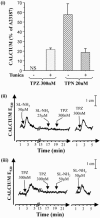
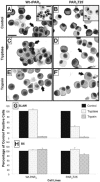
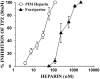
1. Human mast cell tryptase appears to display considerable variation in activating proteinase-activated receptor 2 (PAR(2)). We found tryptase to be an inefficient activator of wild-type rat-PAR(2) (wt-rPAR(2)) and therefore decided to explore the factors that may influence tryptase activation of PAR(2). 2. Using a 20 mer peptide (P20) corresponding to the cleavage/activation sequence of wt-rPAR(2), tryptase was as efficient as trypsin in releasing the receptor-activating sequence (SLIGRL.). However, in the presence of either human-PAR(2) or wt-r PAR(2) expressing cells, tryptase could only activate PAR(2) by releasing SLIGRL from the P20 peptide, suggesting that PAR(2) expressed on the cells was protected from tryptase activation. 3. Three approaches were employed to test the hypothesis that PAR(2) receptor glycosylation restricts tryptase activation. (a) pretreatment of wt-rPAR(2) expressing cells or human embryonic kidney cells (HEK293) with vibrio cholerae neuraminidase to remove oligosaccharide sialic acid, unmasked tryptase-mediated PAR(2) activation. (b) Inhibiting receptor glycosylation in HEK293 cells with tunicamycin enabled tryptase-mediated PAR(2) activation. (c) Wt-rPAR(2) devoid of the N-terminal glycosylation sequon (PAR(2)T25(-)), but not rPAR(2) devoid of the glycosylation sequon located on extracellular loop-2 (PAR(2)T224A), was selectively and substantially (>30 fold) more sensitive to tryptase compared with the wt-rPAR(2). 4. Immunocytochemistry using antisera that specifically recognized the N-terminal precleavage sequence of PAR(2) demonstrated that tryptase released the precleavage domain from PAR(2)T25(-) but not from wt-rPAR(2). 5. Heparin : tryptase molar ratios of greater than 2 : 1 abrogated tryptase activation of PAR(2)T25(-). 6. Our results indicate that glycosylation of PAR(2) and heparin-inhibition of PAR(2) activation by tryptase could provide novel mechanisms for regulating receptor activation by tryptase and possibly other proteases.
Figures

References
-
- AL ANI B., SAIFEDDINE M., KAWABATA A., RENAUX B., MOKASHI S., HOLLENBERG M.D. Proteinase-activated receptor 2 (PAR(2)): development of a ligand- binding assay correlating with activation of PAR(2) by PAR(1)- and PAR(2)-derived peptide ligands. J. Pharmacol. Exp. Ther. 1999;290:753–760. - PubMed
-
- ALM A.K., GAGNEMO-PERSSON R., SORSA T., SUNDELIN J. Extrapancreatic trypsin-2 cleaves proteinase-activated receptor-2. Biochem. Biophys. Res. Commun. 2000;275:77–83. - PubMed
-
- BROWN J.K., TYLER C.L., JONES C.A., RUOSS S.J., HARTMANN T., CAUGHEY G.H. Tryptase, the dominant secretory granular protein in human mast cells, is a potent mitogen for cultured dog tracheal smooth muscle cells. Am. J. Respir. Cell Mol. Biol. 1995;13:227–236. - PubMed
-
- CAIRNS J.A., WALLS A.F. Mast cell tryptase is a mitogen for epithelial cells. Stimulation of IL- 8 production and intercellular adhesion molecule-1 expression. J. Immunol. 1996;156:275–283. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

