The cyclin B1 gene is actively transcribed during mitosis in HeLa cells
- PMID: 11606417
- PMCID: PMC1084122
- DOI: 10.1093/embo-reports/kve223
The cyclin B1 gene is actively transcribed during mitosis in HeLa cells
Abstract
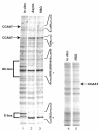
In mammalian cells, the expression level of the cyclin B1 gene plays a critical role in the progression through mitosis. Here we demonstrate that the transcriptional activity of the human cyclin B1 promoter, as well as the rate of gene transcription, is high during mitosis. Indeed, the cyclin B1 promoter maintains an open chromatin configuration at the mitotic stage. Consistent with this, we show that the cyclin B1 promoter is occupied and bound to NF-Y during mitosis in vivo. Our results provide the first example of RNA polymerase II-dependent transcription during mitosis in mammalian cells.
Figures
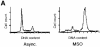
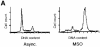




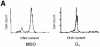

Similar articles
-
NF-Y mediates the transcriptional inhibition of the cyclin B1, cyclin B2, and cdc25C promoters upon induced G2 arrest.J Biol Chem. 2001 Feb 23;276(8):5570-6. doi: 10.1074/jbc.M006052200. Epub 2000 Nov 28. J Biol Chem. 2001. PMID: 11096075
-
p53 is a NF-Y- and p21-independent, Sp1-dependent repressor of cyclin B1 transcription.FEBS Lett. 2005 Feb 14;579(5):1001-7. doi: 10.1016/j.febslet.2004.12.073. Epub 2005 Jan 13. FEBS Lett. 2005. PMID: 15710382
-
Inhibition of CBF/NF-Y mediated transcription activation arrests cells at G2/M phase and suppresses expression of genes activated at G2/M phase of the cell cycle.Nucleic Acids Res. 2006;34(21):6272-85. doi: 10.1093/nar/gkl801. Epub 2006 Nov 10. Nucleic Acids Res. 2006. PMID: 17098936 Free PMC article.
-
Three CCAAT-boxes and a single cell cycle genes homology region (CHR) are the major regulating sites for transcription from the human cyclin B2 promoter.Gene. 2003 Jul 17;312:225-37. doi: 10.1016/s0378-1119(03)00618-8. Gene. 2003. PMID: 12909359
-
Cyclin B1 expression in response to abrogation of the radiation-induced G2/M block in HeLa cells.Cell Prolif. 1998 Apr;31(2):49-57. doi: 10.1046/j.1365-2184.1998.00107.x. Cell Prolif. 1998. PMID: 9745615 Free PMC article.
Cited by
-
Glucose restriction induces cell death in parental but not in homeodomain-interacting protein kinase 2-depleted RKO colon cancer cells: molecular mechanisms and implications for tumor therapy.Cell Death Dis. 2013 May 23;4(5):e639. doi: 10.1038/cddis.2013.163. Cell Death Dis. 2013. PMID: 23703384 Free PMC article.
-
The HIPK2/CDC14B-MeCP2 axis enhances the spindle assembly checkpoint block by promoting cyclin B translation.Sci Adv. 2023 Jan 20;9(3):eadd6982. doi: 10.1126/sciadv.add6982. Epub 2023 Jan 20. Sci Adv. 2023. PMID: 36662865 Free PMC article.
-
Importance of Sox2 in maintenance of cell proliferation and multipotency of mesenchymal stem cells in low-density culture.Cell Prolif. 2011 Oct;44(5):428-40. doi: 10.1111/j.1365-2184.2011.00770.x. Cell Prolif. 2011. PMID: 21951286 Free PMC article.
-
Molecular imaging of nuclear factor-Y transcriptional activity maps proliferation sites in live animals.Mol Biol Cell. 2012 Apr;23(8):1467-74. doi: 10.1091/mbc.E12-01-0039. Epub 2012 Feb 29. Mol Biol Cell. 2012. PMID: 22379106 Free PMC article.
-
Sirtuins-Mediated System-Level Regulation of Mammalian Tissues at the Interface between Metabolism and Cell Cycle: A Systematic Review.Biology (Basel). 2021 Mar 4;10(3):194. doi: 10.3390/biology10030194. Biology (Basel). 2021. PMID: 33806509 Free PMC article. Review.
References
-
- Clute P. and Pines, J. (1999) Temporal and spatial control of cyclin B1 destruction in metaphase. Nature Cell. Biol., 1, 82–87. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

