Titration of an SIVmac251 stock by vaginal inoculation of Indian and Chinese origin rhesus macaques: transmission efficiency, viral loads, and antibody responses
- PMID: 11679158
- PMCID: PMC3401017
- DOI: 10.1089/088922201753197123
Titration of an SIVmac251 stock by vaginal inoculation of Indian and Chinese origin rhesus macaques: transmission efficiency, viral loads, and antibody responses
Abstract
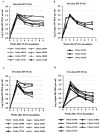
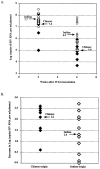
The purpose of this study was to determine whether rhesus monkeys of Chinese origin are suitable for studies of mucosal lentivirus transmission by comparing the relative ability of these animals and rhesus macaques of Indian origin to become infected by vaginal (IVAG) inoculation with SIVmac251. In addition, we sought to test the hypothesis that differences in viral load during the first few weeks after inoculation were due to the relative strength of the anti-SIV immune responses in the two populations of rhesus macaques. Significant difference was not observed between the number of Indian and Chinese origin monkeys that were infected after IVAG SIV inoculation in this study. For 8-9 weeks after infection there was considerable overlap in the range of viral loads among the Indian and Chinese animals and the variation among the Indian origin animals was greater than the variation among the Chinese origin monkeys. By 6 weeks postinfection, viral loads in SIV-infected Chinese origin monkeys tended to be at the lower end of the range of viral loads observed in SIV-infected Indian origin monkeys. The strength of the anti-SIV antibody response was also more variable in the Indian origin rhesus macaques, but at 6-8 weeks postinfection, Chinese and Indian origin rhesus macaques had similar titers of anti-SIV antibodies. Microsatellite allele frequencies differed between Chinese and Indian rhesus macaques; however, the majority of alleles present in Indian-origin animals were also found in Chinese macaques. Together these results show that host factors, other than geographic origin, determine the ability of a rhesus macaque to be infected after IVAG SIV exposure and that geographic origin does not predict the viral load of SIV-infected animals during the first 8-9 weeks after IVAG inoculation.
Figures
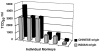
Similar articles
-
Route of simian immunodeficiency virus inoculation determines the complexity but not the identity of viral variant populations that infect rhesus macaques.J Virol. 2001 Apr;75(8):3753-65. doi: 10.1128/JVI.75.8.3753-3765.2001. J Virol. 2001. PMID: 11264364 Free PMC article.
-
SIV(mac) pathogenesis in rhesus macaques of Chinese and Indian origin compared with primary HIV infections in humans.AIDS. 2002 Jul 26;16(11):1489-96. doi: 10.1097/00002030-200207260-00005. AIDS. 2002. PMID: 12131186
-
Passive infusion of immune serum into simian immunodeficiency virus-infected rhesus macaques undergoing a rapid disease course has minimal effect on plasma viremia.Virology. 2000 Apr 25;270(1):237-49. doi: 10.1006/viro.2000.0254. Virology. 2000. PMID: 10772996
-
Localization of Simian immunodeficiency virus-infected cells in the genital tract of male and female Rhesus macaques.J Reprod Immunol. 1998 Dec;41(1-2):331-9. doi: 10.1016/s0165-0378(98)00069-2. J Reprod Immunol. 1998. PMID: 10213321 Review.
-
The role of disease stage, plasma viral load and regulatory T cells (Tregs) on autoantibody production in SIV-infected non-human primates.J Autoimmun. 2007 Mar-May;28(2-3):152-9. doi: 10.1016/j.jaut.2007.02.014. Epub 2007 Mar 26. J Autoimmun. 2007. PMID: 17368846 Free PMC article. Review.
Cited by
-
Pathogenicity of simian-human immunodeficiency virus SHIV-89.6P and SIVmac is attenuated in cynomolgus macaques and associated with early T-lymphocyte responses.J Virol. 2005 Jul;79(14):8878-85. doi: 10.1128/JVI.79.14.8878-8885.2005. J Virol. 2005. PMID: 15994781 Free PMC article.
-
SIV infection of rhesus macaques of Chinese origin: a suitable model for HIV infection in humans.Retrovirology. 2013 Aug 15;10:89. doi: 10.1186/1742-4690-10-89. Retrovirology. 2013. PMID: 23947613 Free PMC article. Review.
-
Early detection of simian immunodeficiency virus in the central nervous system following oral administration to rhesus macaques.Front Immunol. 2013 Aug 14;4:236. doi: 10.3389/fimmu.2013.00236. eCollection 2013. Front Immunol. 2013. PMID: 23966995 Free PMC article.
-
SIVmac251 is inefficiently transmitted to rhesus macaques by penile inoculation with a single SIVenv variant found in ramp-up phase plasma.AIDS Res Hum Retroviruses. 2011 Dec;27(12):1259-69. doi: 10.1089/aid.2011.0090. Epub 2011 Jul 6. AIDS Res Hum Retroviruses. 2011. PMID: 21732792 Free PMC article.
-
Identification of Unequally Represented Founder Viruses Among Tissues in Very Early SIV Rectal Transmission.Front Microbiol. 2018 Mar 29;9:557. doi: 10.3389/fmicb.2018.00557. eCollection 2018. Front Microbiol. 2018. PMID: 29651274 Free PMC article.
References
-
- Joag SV, Stephens EB, Adams RJ, Foresman L, Narayan O. Pathogenesis of SIVmac infection in Chinese and Indian rhesus macaques: Effects of splenectomy on virus burden. Virology. 1994;200:436–446. - PubMed
-
- Committee on Care and Use of Laboratory Animals of the Institute of Laboratory Resources. Guide for Care and Use of Laboratory Animals. National Resource Council; Washington, D.C.: 1998.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

