The lung as a route for systemic delivery of therapeutic proteins and peptides
- PMID: 11686885
- PMCID: PMC59577
- DOI: 10.1186/rr58
The lung as a route for systemic delivery of therapeutic proteins and peptides
Abstract
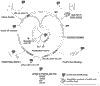
The large surface area, good vascularization, immense capacity for solute exchange and ultra-thinness of the alveolar epithelium are unique features of the lung that can facilitate systemic delivery via pulmonary administration of peptides and proteins. Physical and biochemical barriers, lack of optimal dosage forms and delivery devices limit the systemic delivery of biotherapeutic agents by inhalation. Current efforts to overcome these difficulties in order to deliver metabolic hormones (insulin, calcitonin, thyroid-stimulating hormone [TSH], follicle-stimulating hormone [FSH] and growth hormones) systemically, to induce systemic responses (immunoglobulins, cyclosporin A [CsA], recombinant-methionyl human granulocyte colony-stimulating factor [r-huG-CSF], pancreatic islet autoantigen) and to modulate other biological processes via the lung are reviewed. Safety aspects of pulmonary peptide and protein administration are also discussed.
Figures
References
-
- Wearley LL. Recent progress in protein and peptide delivery by non-invasive routes. Crit Rev Ther Drug Carrier Syst. 1991;8:1331–1394. - PubMed
-
- Wilson CG, Washington N. Physiological Pharmaceutics England: John Wiley; 1989.
-
- Banga AK. Therapeutic Peptides and Proteins: Formulation, Processing and Delivery Systems Lancaster: Technomic; 1995.
-
- Weibel ER. Morphometry of the Human Lung Berlin: Springer Verlag; 1963.
-
- Chrystyn H. Is total particle dose more important than particle distribution. Respir Med. 1997;91(suppl):17–19. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

