Vaccinia virus infection disarms the mitochondrion-mediated pathway of the apoptotic cascade by modulating the permeability transition pore
- PMID: 11689625
- PMCID: PMC114730
- DOI: 10.1128/JVI.75.23.11437-11448.2001
Vaccinia virus infection disarms the mitochondrion-mediated pathway of the apoptotic cascade by modulating the permeability transition pore
Abstract
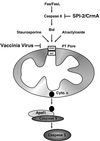
Many viruses have evolved strategies that target crucial components within the apoptotic cascade. One of the best studied is the caspase 8 inhibitor, crmA/Spi-2, encoded by members of the poxvirus family. Since many proapoptotic stimuli induce apoptosis through a mitochondrion-dependent, caspase 8-independent pathway, we hypothesized that vaccinia virus would encode a mechanism to directly modulate the mitochondrial apoptotic pathway. In support of this, we observed that Jurkat cells, which undergo Fas-mediated apoptosis exclusively through the mitochondrial route, were resistant to Fas-induced death following infection with a crmA/Spi-2-deficient strain of vaccinia virus. In addition, vaccinia virus-infected cells subjected to the proapoptotic stimulus staurosporine exhibited decreased levels of both cytochrome c released from the mitochondria and caspase 3 activation. In all cases we found that the loss of the mitochondrial membrane potential, which occurs as a result of opening the multimeric permeability transition pore complex, was prevented in vaccinia virus-infected cells. Moreover, vaccinia virus infection specifically inhibited opening of the permeability transition pore following treatment with the permeability transition pore ligand atractyloside and t-butylhydroperoxide. These studies indicate that vaccinia virus infection directly impacts the mitochondrial apoptotic cascade by influencing the permeability transition pore.
Figures





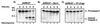
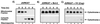


Similar articles
-
The vaccinia virus F1L protein interacts with the proapoptotic protein Bak and inhibits Bak activation.J Virol. 2005 Nov;79(22):14031-43. doi: 10.1128/JVI.79.22.14031-14043.2005. J Virol. 2005. PMID: 16254338 Free PMC article.
-
Tumor-induced apoptosis of T cells: amplification by a mitochondrial cascade.Cancer Res. 2000 Dec 15;60(24):6811-7. Cancer Res. 2000. PMID: 11156370
-
Vaccinia virus encodes a previously uncharacterized mitochondrial-associated inhibitor of apoptosis.Proc Natl Acad Sci U S A. 2003 Nov 25;100(24):14345-50. doi: 10.1073/pnas.2235583100. Epub 2003 Nov 10. Proc Natl Acad Sci U S A. 2003. PMID: 14610284 Free PMC article.
-
And all of a sudden it's over: mitochondrial outer-membrane permeabilization in apoptosis.Biochimie. 2002 Feb-Mar;84(2-3):113-21. doi: 10.1016/s0300-9084(02)01379-2. Biochimie. 2002. PMID: 12022942 Review.
-
Mitochondrial regulation of apoptosis.News Physiol Sci. 2003 Jun;18:89-94. doi: 10.1152/nips.01433.2002. News Physiol Sci. 2003. PMID: 12750442 Review.
Cited by
-
The vaccinia virus F1L protein interacts with the proapoptotic protein Bak and inhibits Bak activation.J Virol. 2005 Nov;79(22):14031-43. doi: 10.1128/JVI.79.22.14031-14043.2005. J Virol. 2005. PMID: 16254338 Free PMC article.
-
Sheeppox virus SPPV14 encodes a Bcl-2-like cell death inhibitor that counters a distinct set of mammalian proapoptotic proteins.J Virol. 2012 Nov;86(21):11501-11. doi: 10.1128/JVI.01115-12. Epub 2012 Aug 15. J Virol. 2012. PMID: 22896610 Free PMC article.
-
Tumorigenic poxviruses up-regulate intracellular superoxide to inhibit apoptosis and promote cell proliferation.J Virol. 2005 May;79(9):5799-811. doi: 10.1128/JVI.79.9.5799-5811.2005. J Virol. 2005. PMID: 15827194 Free PMC article.
-
Vaccinia virus B1R kinase interacts with JIP1 and modulates c-Jun-dependent signaling.J Virol. 2006 Aug;80(15):7667-75. doi: 10.1128/JVI.00967-06. J Virol. 2006. PMID: 16840345 Free PMC article.
-
Vaccinia virus F1L interacts with Bak using highly divergent Bcl-2 homology domains and replaces the function of Mcl-1.J Biol Chem. 2010 Feb 12;285(7):4695-708. doi: 10.1074/jbc.M109.053769. Epub 2009 Dec 2. J Biol Chem. 2010. PMID: 19955184 Free PMC article.
References
-
- Barry M, McFadden G. Apoptosis regulators from DNA viruses. Curr Opin Immunol. 1998;10:422–430. - PubMed
-
- Barry M, Heibein J, Pinkoski M, Bleackley R C. Quantitative measurement of apoptosis induced by cytotoxic T lymphocytes. Methods Enzymol. 2000;322:40–46. - PubMed
-
- Bertin J, Armstrong R C, Ottilie S, Martin D A, Wang Y, Banks S, Wang G H, Senkevich T G, Alnemri E M, Moss B, Lenardo M J, Tomaselli K J, Cohen J I. Death effector domain-containing herpesvirus and poxvirus proteins inhibit both Fas- and TNFR1-induced apoptosis. Proc Natl Acad Sci USA. 1997;94:1772–1776. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

