Cell density and phosphorylation control the subcellular localization of adenomatous polyposis coli protein
- PMID: 11689703
- PMCID: PMC99979
- DOI: 10.1128/MCB.21.23.8143-8156.2001
Cell density and phosphorylation control the subcellular localization of adenomatous polyposis coli protein
Abstract
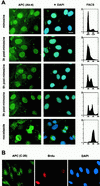
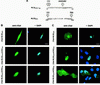
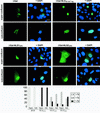
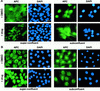
Loss of functional adenomatous polyposis coli protein (APC) leads to uncontrolled proliferation of colonic epithelial cells, as evidenced by polyp formation, a prelude to carcinogenesis. As a tumor suppressor, APC targets the oncogene beta-catenin for proteasome-mediated cytoplasmic degradation. Recently, it was demonstrated that APC also interacts with nuclear beta-catenin, thereby reducing beta-catenin's activity as a transcription cofactor and enhancing its nuclear export. The first objective of this study was to analyze how cellular context affected APC distribution. We determined that cell density but not cell cycle influenced APC's subcellular distribution, with predominantly nuclear APC found in subconfluent MDCK and intestinal epithelial cells but both cytoplasmic and nuclear APC in superconfluent cells. Redistribution of APC protein did not depend on continual nuclear export. Focusing on the two defined nuclear localization signals in the C-terminal third of APC (NLS1(APC) and NLS2(APC)), we found that phosphorylation at the CK2 site increased and phosphorylation at the PKA site decreased NLS2(APC)-mediated nuclear translocation. Cell density-mediated redistribution of beta-galactosidase was achieved by fusion to NLS2(APC) but not to NLS1(APC). Both the CK2 and PKA sites were important for this density-mediated redistribution, and pharmacological agents that target CK2 and PKA instigated relocalization of endogenous APC. Our data provide evidence that physiological signals such as cell density regulate APC's nuclear distribution, with phosphorylation sites near NLS2(APC) being critical for this regulation.
Figures





References
-
- Aukema H M, Davidson L A, Pence B C, Jiang Y H, Lupton J R, Chapkin R S. Butyrate alters activity of specific cAMP-receptor proteins in a transgenic mouse colonic cell line. J Nutr. 1997;127:18–24. - PubMed
-
- Behrens J, Jerchow B A, Wurtele M, Grimm J, Asbrand C, Wirtz R, Kuhl M, Wedlich D, Birchmeier W. Functional interaction of an axin homolog, conductin, with β-catenin, APC, and GSK3β. Science. 1998;280:596–599. - PubMed
-
- Conti E, Uy M, Leighton L, Blobel G, Kuriyan J. Crystallographic analysis of the recognition of a nuclear localization signal by the nuclear import factor karyopherin-α. Cell. 1998;94:193–204. - PubMed
-
- Dingwall C, Laskey R A. Nuclear targeting sequences–a consensus? Trends Biochem Sci. 1991;16:478–481. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
