Senescence is induced in individually darkened Arabidopsis leaves, but inhibited in whole darkened plants
- PMID: 11706170
- PMCID: PMC129259
Senescence is induced in individually darkened Arabidopsis leaves, but inhibited in whole darkened plants
Abstract
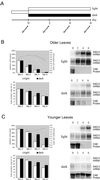
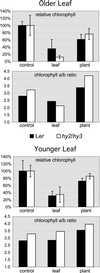
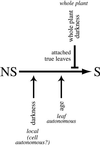
It has long been known that leaf senescence can be induced in many plant species by detaching leaves and placing them in the darkness. It recently has been shown that entire Arabidopsis plants placed in the darkness are not induced to senesce, as judged by visible yellowing and certain molecular markers. Here, we show that when individual Arabidopsis leaves are darkened, but not when entire plants are darkened, senescence is induced in the covered leaves. This induction of senescence is highly localized. The phenomenon is leaf age dependent in that it occurs more rapidly and strongly in older leaves than in younger ones, as is the case with many forms of induced senescence. Whole adult plants placed in darkness, in contrast, show delayed senescence, although seedlings lacking primary leaves do not. These observations imply that the light status of the entire plant affects the senescence of individual leaves. A model summarizing the results is presented.
Figures



References
-
- Biswal UC, Biswal B. Photocontrol of leaf senescence. Photochem Photobiol. 1984;39:875–879.
-
- Biswal B, Choudhury NK. Photocontrol of chlorophyll loss in papaya leaf discs. Plant Cell Physiol. 1986;27:1439–1444.
-
- Biswal B, Choudhury NK, Sahu P, Biswal UC. Senescence of detached fern leaves. Plant Cell Physiol. 1983;24:1203–1208.
-
- Biswal UC, Kasemir H, Mohr H. Phytochrome control of degreening of attached cotyledons and primary leaves of mustard (Sinapis alba L.) seedlings. Photochem Photobiol. 1982;35:237–241.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
