p38 MAP kinase modulates liver cell volume through inhibition of membrane Na+ permeability
- PMID: 11714741
- PMCID: PMC209415
- DOI: 10.1172/JCI12190
p38 MAP kinase modulates liver cell volume through inhibition of membrane Na+ permeability
Abstract
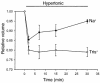
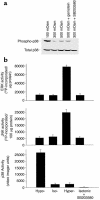
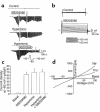
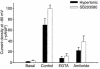
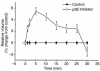
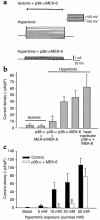
In hepatocytes, Na+ influx through nonselective cation (NSC) channels represents a key point for regulation of cell volume. Under basal conditions, channels are closed, but both physiologic and pathologic stimuli lead to a large increase in Na+ and water influx. Since osmotic stimuli also activate mitogen-activated protein (MAP) kinase pathways, we have examined regulation of Na+ permeability and cell volume by MAP kinases in an HTC liver cell model. Under isotonic conditions, there was constitutive activity of p38 MAP kinase that was selectively inhibited by SB203580. Decreases in cell volume caused by hypertonic exposure had no effect on p38, but increases in cell volume caused by hypotonic exposure increased p38 activity tenfold. Na+ currents were small when cells were in isotonic media but could be increased by inhibiting constitutive p38 MAP kinase, thereby increasing cell volume. To evaluate the potential inhibitory role of p38 more directly, cells were dialyzed with recombinant p38alpha and its upstream activator, MEK-6, which substantially inhibited volume-sensitive currents. These findings indicate that constitutive p38 activity contributes to the low Na+ permeability necessary for maintenance of cell volume, and that recombinant p38 negatively modulates the set point for volume-sensitive channel opening. Thus, functional interactions between p38 MAP kinase and ion channels may represent an important target for modifying volume-sensitive liver functions.
Figures

References
-
- Paul A, et al. Stress-activated protein kinases: activation, regulation and function. Cell Signal. 1997;9:403–410. - PubMed
-
- Bode JG, et al. The mitogen-activated protein (MAP) kinase p38 and its upstream activator MAP kinase kinase 6 are involved in the activation of signal transducer and activator of transcription by hyperosmolarity. J Biol Chem. 1999;274:30222–30227. - PubMed
-
- Han J, Richter B, Li Z, Kravchenko V, Ulevitch RJ. Molecular cloning of human p38 MAP kinase. . Biochim Biophys Acta. 1995; 1265:224–227. - PubMed
-
- Han J, Lee JD, Bibbs L, Ulevitch RJ. A MAP kinase targeted by endotoxin and hyperosmolarity in mammalian cells. Science. 1994;265:808–811. - PubMed
-
- Derijard B, et al. Independent human MAP-kinase signal transduction pathways defined by MEK and MKK isoforms [erratum 1995, 269(5220):17] Science. 1995;267:682–685. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

