Structure determination of human and murine beta-defensins reveals structural conservation in the absence of significant sequence similarity
- PMID: 11714914
- PMCID: PMC2374044
- DOI: 10.1110/ps.24401
Structure determination of human and murine beta-defensins reveals structural conservation in the absence of significant sequence similarity
Abstract
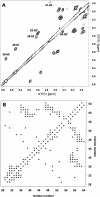
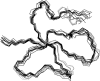
Defensins are cationic and cysteine-rich peptides that play a crucial role in the host defense against microorganisms of many organisms by their capability to permeabilize bacterial membranes. The low sequence similarity among the members of the large mammalian beta-defensin family suggests that their antimicrobial activity is largely independent of their primary structure. To investigate to what extent these defensins share a similar fold, the structures of the two human beta-defensins, hBD-1 and hBD-2, as well as those of two novel murine defensins, termed mBD-7 and mBD-8, were determined by nuclear magnetic resonance spectroscopy. All four defensins investigated share a striking similarity on the level of secondary and tertiary structure including the lack of a distinct hydrophobic core, suggesting that the fold is mainly stabilized by the presence of three disulfide bonds. In addition to the overall shape of the molecules, the ratio of solvent-exposed polar and hydrophobic side chains is also very similar among the four defensins investigated. It is significant that beta-defensins do not exhibit a common pattern of charged and hydrophobic residues on the protein surface and that the beta-defensin-specific fold appears to accommodate a wide range of different amino acids at most sequence positions. In addition to the implications for the mode of biological defensin actions, these findings are of particular interest because beta-defensins have been suggested as lead compounds for the development of novel peptide antibiotics for the therapy of infectious diseases.
Figures


References
-
- Baba, M., Imai, T., Nishimura, M., Kakizaki, M., Takagi, S., Hieshima, K., Nomiyama, H., and Yoshie, O. 1997. Identification of CCR6, the specific receptor for a novel lymphocyte-directed CC chemokine LARC. J. Biol. Chem. 272 14893–14898. - PubMed
-
- Barton, G.J. 1993. ALSCRIPT: A tool to format multiple sequence alignments. Protein Eng. 6 37–40. - PubMed
-
- Bensch, K.W., Raida, M., Mägert, H.J., Schulz-Knappe, P., and Forssmann, W.G. 1995. hBD-1: A novel beta-defensin from human plasma. FEBS Lett. 368 331–335. - PubMed
-
- Boman, H.G. 1995. Peptide antibiotics and their role in innate immunity. Annu. Rev. Immunol. 13 61–92. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

