Role of positively charged transmembrane segments in the insertion and assembly of mitochondrial inner-membrane proteins
- PMID: 11717439
- PMCID: PMC61124
- DOI: 10.1073/pnas.251503098
Role of positively charged transmembrane segments in the insertion and assembly of mitochondrial inner-membrane proteins
Abstract
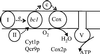
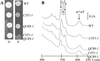
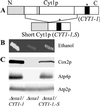
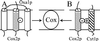
The biogenesis of membrane oligomeric complexes is an intricate process that requires the insertion and assembly of transmembrane (TM) domains into the lipid bilayer. The Oxa1p family plays a key role in this process in organelles and bacteria. Hell et al. (2001, EMBO J., 20, 1281-1288) recently have proposed that Oxa1p could act as part of a general membrane insertion machinery for mitochondrial respiratory complex subunits. We have previously shown that mutations in the TM domain of Cyt1p can partially compensate for the absence of Oxa1p. Here, we demonstrate that a single amino acid substitution in the TM domain of Qcr9p can bypass Oxa1p in yeast. Qcr9p and Cyt1p are two subunits of the respiratory complex bc1 and their relative roles in the assembly of other respiratory complexes have been investigated. The mutations we have isolated in Cyt1p or Qcr9p introduce positively charged amino acids, and we show that the mutant TM domain of Cyt1p mediates the restoration of complex assembly. We propose that the positive charges introduced in Cyt1p and Qcr9p TM domains promote interactions with negatively charged TM domains of other respiratory complex subunits, allowing the coinsertion of both domains into the membrane, in the absence of Oxa1p. This model argues in favor of a role of Oxa1p in the insertion and the lateral exit of less hydrophobic TM domains from the translocation site into the lipid bilayer.
Figures



Similar articles
-
Rmd9p controls the processing/stability of mitochondrial mRNAs and its overexpression compensates for a partial deficiency of oxa1p in Saccharomyces cerevisiae.Genetics. 2007 Mar;175(3):1105-15. doi: 10.1534/genetics.106.063883. Epub 2006 Dec 28. Genetics. 2007. PMID: 17194787 Free PMC article.
-
Insertion into the mitochondrial inner membrane of a polytopic protein, the nuclear-encoded Oxa1p.EMBO J. 1997 May 1;16(9):2217-26. doi: 10.1093/emboj/16.9.2217. EMBO J. 1997. PMID: 9171337 Free PMC article.
-
Oxa1p acts as a general membrane insertion machinery for proteins encoded by mitochondrial DNA.EMBO J. 2001 Mar 15;20(6):1281-8. doi: 10.1093/emboj/20.6.1281. EMBO J. 2001. PMID: 11250894 Free PMC article.
-
Inserting membrane proteins: the YidC/Oxa1/Alb3 machinery in bacteria, mitochondria, and chloroplasts.Biochim Biophys Acta. 2011 Mar;1808(3):866-75. doi: 10.1016/j.bbamem.2010.08.014. Epub 2010 Aug 26. Biochim Biophys Acta. 2011. PMID: 20800571 Review.
-
YidC/Oxa1p/Alb3: evolutionarily conserved mediators of membrane protein assembly.FEBS Lett. 2001 Jul 13;501(1):1-5. doi: 10.1016/s0014-5793(01)02616-3. FEBS Lett. 2001. PMID: 11457446 Review.
Cited by
-
Ribosome binding to the Oxa1 complex facilitates co-translational protein insertion in mitochondria.EMBO J. 2003 Dec 15;22(24):6448-57. doi: 10.1093/emboj/cdg623. EMBO J. 2003. PMID: 14657018 Free PMC article.
-
Conserved negative charges in the transmembrane segments of subunit K of the NADH:ubiquinone oxidoreductase determine its dependence on YidC for membrane insertion.J Biol Chem. 2010 Feb 5;285(6):3575-3581. doi: 10.1074/jbc.M109.051128. Epub 2009 Dec 3. J Biol Chem. 2010. PMID: 19959836 Free PMC article.
-
The role of the 3' untranslated region in mRNA sorting to the vicinity of mitochondria is conserved from yeast to human cells.Mol Biol Cell. 2003 Sep;14(9):3848-56. doi: 10.1091/mbc.e03-02-0074. Epub 2003 Jun 13. Mol Biol Cell. 2003. PMID: 12972568 Free PMC article.
-
Hydrophilic microenvironment required for the channel-independent insertase function of YidC protein.Proc Natl Acad Sci U S A. 2015 Apr 21;112(16):5063-8. doi: 10.1073/pnas.1423817112. Epub 2015 Apr 8. Proc Natl Acad Sci U S A. 2015. PMID: 25855636 Free PMC article.
-
Rmd9p controls the processing/stability of mitochondrial mRNAs and its overexpression compensates for a partial deficiency of oxa1p in Saccharomyces cerevisiae.Genetics. 2007 Mar;175(3):1105-15. doi: 10.1534/genetics.106.063883. Epub 2006 Dec 28. Genetics. 2007. PMID: 17194787 Free PMC article.
References
-
- de Gier J-W, Luirink J. Mol Microbiol. 2001;40:314–322. - PubMed
-
- Rapoport T A, Jungnickel B, Kutay U. Annu Rev Biochem. 1996;65:271–303. - PubMed
-
- Grivell L A. Crit Rev Biochem Mol Biol. 1995;30:121–164. - PubMed
-
- Herrmann J, Neupert W. Curr Opin Microbiol. 2000;3:210–214. - PubMed
-
- Tokatlidis K, Schatz G. J Biol Chem. 1999;274:35285–35288. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

