p27 cytoplasmic localization is regulated by phosphorylation on Ser10 and is not a prerequisite for its proteolysis
- PMID: 11726503
- PMCID: PMC125773
- DOI: 10.1093/emboj/20.23.6672
p27 cytoplasmic localization is regulated by phosphorylation on Ser10 and is not a prerequisite for its proteolysis
Abstract
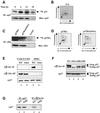
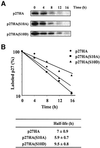
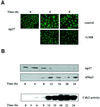
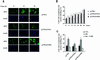
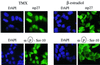
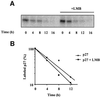
The activity of the cyclin-dependent kinase inhibitor p27 is controlled by its concentration and subcellular localization. However, the mechanisms that regulate its intracellular transport are poorly understood. Here we show that p27 is phosphorylated on Ser10 in vivo and that mutation of Ser10 to Ala inhibits p27 cytoplasmic relocalization in response to mitogenic stimulation. In contrast, a fraction of wild-type p27 and a p27(S10D)-phospho-mimetic mutant translocates to the cytoplasm in the presence of mitogens. G1 nuclear export of p27 and its Ser10 phosphorylation precede cyclin-dependent kinase 2 (Cdk2) activation and degradation of the bulk of p27. Interestingly, leptomycin B-mediated nuclear accumulation accelerates the turnover of endogenous p27; the p27(S10A) mutant, which is trapped in the nucleus, has a shorter half-life than wild-type p27 and the p27(S10D) mutant. In summary, p27 is efficiently degraded in the nucleus and phosphorylation of Ser10 is necessary for the nuclear to cytoplasmic redistribution of a fraction of p27 in response to mitogenic stimulation. This cytoplasmic localization may serve to decrease the abundance of p27 in the nucleus below a certain threshold required for activation of cyclin-Cdk2 complexes.
Figures



Similar articles
-
Estrogens down-regulate p27Kip1 in breast cancer cells through Skp2 and through nuclear export mediated by the ERK pathway.J Biol Chem. 2003 Oct 17;278(42):41355-66. doi: 10.1074/jbc.M302830200. Epub 2003 Aug 6. J Biol Chem. 2003. PMID: 12904306
-
Phosphorylation of p27Kip1 on serine 10 is required for its binding to CRM1 and nuclear export.J Biol Chem. 2002 Apr 26;277(17):14355-8. doi: 10.1074/jbc.C100762200. Epub 2002 Mar 11. J Biol Chem. 2002. PMID: 11889117
-
Mutations of phosphorylation sites Ser10 and Thr187 of p27Kip1 abolish cytoplasmic redistribution but do not abrogate G0/1 phase arrest in the HepG2 cell line.Biochem Biophys Res Commun. 2006 Sep 1;347(3):601-7. doi: 10.1016/j.bbrc.2006.06.114. Epub 2006 Jun 28. Biochem Biophys Res Commun. 2006. PMID: 16842750
-
Role of serine 10 phosphorylation in p27 stabilization revealed by analysis of p27 knock-in mice harboring a serine 10 mutation.J Biol Chem. 2005 Jan 14;280(2):1095-102. doi: 10.1074/jbc.M406117200. Epub 2004 Nov 3. J Biol Chem. 2005. PMID: 15528185
-
Cytoplasmic displacement of cyclin E-cdk2 inhibitors p21Cip1 and p27Kip1 in anchorage-independent cells.Oncogene. 1998 May;16(20):2575-83. doi: 10.1038/sj.onc.1201791. Oncogene. 1998. PMID: 9632134
Cited by
-
Hypoxia inducible prolyl hydroxylase PHD3 maintains carcinoma cell growth by decreasing the stability of p27.Mol Cancer. 2015 Jul 30;14:143. doi: 10.1186/s12943-015-0410-5. Mol Cancer. 2015. PMID: 26223520 Free PMC article.
-
Dual effects of Ral-activated pathways on p27 localization and TGF-β signaling.Mol Biol Cell. 2013 Jun;24(11):1812-24. doi: 10.1091/mbc.E13-01-0007. Epub 2013 Apr 10. Mol Biol Cell. 2013. PMID: 23576547 Free PMC article.
-
A growth factor-dependent nuclear kinase phosphorylates p27(Kip1) and regulates cell cycle progression.EMBO J. 2002 Jul 1;21(13):3390-401. doi: 10.1093/emboj/cdf343. EMBO J. 2002. PMID: 12093740 Free PMC article.
-
p27Kip1 modulates cell migration through the regulation of RhoA activation.Genes Dev. 2004 Apr 15;18(8):862-76. doi: 10.1101/gad.1185504. Epub 2004 Apr 12. Genes Dev. 2004. PMID: 15078817 Free PMC article.
-
Characterization of a naturally-occurring p27 mutation predisposing to multiple endocrine tumors.Mol Cancer. 2010 May 21;9:116. doi: 10.1186/1476-4598-9-116. Mol Cancer. 2010. PMID: 20492666 Free PMC article.
References
-
- Boussiotis V.A., Freeman,G.J., Taylor,P.A., Berezovskaya,A., Grass,I., Blazar,B.R. and Nadler,L.M. (2000) p27kip1 functions as an anergy factor inhibiting interleukin 2 transcription and clonal expansion of alloreactive human and mouse helper T lymphocytes. Nature Med., 6, 290–297. - PubMed
-
- Carrano A.C., Eytan,E., Hershko,A. and Pagano,M. (1999) SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nature Cell Biol., 1, 193–199. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

