Cooperation between C/EBPalpha TBP/TFIIB and SWI/SNF recruiting domains is required for adipocyte differentiation
- PMID: 11731483
- PMCID: PMC312836
- DOI: 10.1101/gad.209901
Cooperation between C/EBPalpha TBP/TFIIB and SWI/SNF recruiting domains is required for adipocyte differentiation
Abstract
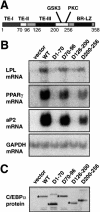
Chromatin remodeling is an important step in promoter activation during cellular lineage commitment and differentiation. We show that the ability of the C/EBPalpha transcription factor to direct adipocyte differentiation of uncommitted fibroblast precursors and to activate SWI/SNF-dependent myeloid-specific genes depends on a domain, C/EBPalpha transactivation element III (TE-III), that binds the SWI/SNF chromatin remodeling complex. TE-III collaborates with C/EBPalpha TBP/TFIIB interaction motifs during induction of adipogenesis and adipocyte-specific gene expression. These results indicate that C/EBPalpha acts as a lineage-instructive transcription factor through SWI/SNF-dependent modification of the chromatin structure of lineage-specific genes, followed by direct promoter activation via recruitment of the basal transcription-initiation complex, and provide a mechanism by which C/EBPalpha can mediate differentiation along multiple cellular lineages.
Figures




Similar articles
-
Sequential gene promoter interactions by C/EBPbeta, C/EBPalpha, and PPARgamma during adipogenesis.Biochem Biophys Res Commun. 2004 May 21;318(1):213-8. doi: 10.1016/j.bbrc.2004.04.017. Biochem Biophys Res Commun. 2004. PMID: 15110775
-
Sequential gene promoter interactions of C/EBPbeta, C/EBPalpha, and PPARgamma during adipogenesis.Biochem Biophys Res Commun. 2004 Jun 18;319(1):235-9. doi: 10.1016/j.bbrc.2004.04.176. Biochem Biophys Res Commun. 2004. PMID: 15158467
-
Mammalian chromatin remodeling complex SWI/SNF is essential for enhanced expression of the albumin gene during liver development.J Biochem. 2006 Feb;139(2):177-88. doi: 10.1093/jb/mvj015. J Biochem. 2006. PMID: 16452305
-
Role of the CCAAT enhancer binding proteins (C/EBPs) in adipocyte differentiation.Biochem Biophys Res Commun. 1999 Dec 29;266(3):677-83. doi: 10.1006/bbrc.1999.1885. Biochem Biophys Res Commun. 1999. PMID: 10603305 Review.
-
ATP-dependent chromatin remodelling: SWI/SNF and Co. are on the job.J Mol Biol. 1999 Oct 22;293(2):187-98. doi: 10.1006/jmbi.1999.2999. J Mol Biol. 1999. PMID: 10529347 Review.
Cited by
-
Modulation of the transcriptional activity of peroxisome proliferator-activated receptor gamma by protein-protein interactions and post-translational modifications.Yonsei Med J. 2013 May 1;54(3):545-59. doi: 10.3349/ymj.2013.54.3.545. Yonsei Med J. 2013. PMID: 23549795 Free PMC article. Review.
-
Brd2 gene disruption causes "metabolically healthy" obesity: epigenetic and chromatin-based mechanisms that uncouple obesity from type 2 diabetes.Vitam Horm. 2013;91:49-75. doi: 10.1016/B978-0-12-407766-9.00003-1. Vitam Horm. 2013. PMID: 23374712 Free PMC article. Review.
-
Histone H3 tail positioning and acetylation by the c-Myb but not the v-Myb DNA-binding SANT domain.Genes Dev. 2005 Oct 15;19(20):2447-57. doi: 10.1101/gad.355405. Epub 2005 Sep 29. Genes Dev. 2005. PMID: 16195416 Free PMC article.
-
MyoD targets chromatin remodeling complexes to the myogenin locus prior to forming a stable DNA-bound complex.Mol Cell Biol. 2005 May;25(10):3997-4009. doi: 10.1128/MCB.25.10.3997-4009.2005. Mol Cell Biol. 2005. PMID: 15870273 Free PMC article.
-
Requirement for SWI/SNF chromatin-remodeling complex in Tat-mediated activation of the HIV-1 promoter.EMBO J. 2006 Apr 19;25(8):1690-9. doi: 10.1038/sj.emboj.7601074. Epub 2006 Apr 6. EMBO J. 2006. PMID: 16601680 Free PMC article.
References
-
- Armstrong JA, Bieker JJ, Emerson BM. A SWI/SNF-related chromatin remodeling complex, E-RC1, is required for tissue-specific transcriptional regulation by EKLF in vitro. Cell. 1998;95:93–104. - PubMed
-
- Barak Y, Nelson MC, Ong ES, Jones YZ, Ruiz-Lozano P, Chien KR, Koder A, Evans RM. PPARγ is required for placental, cardiac, and adipose tissue development. Mol Cell. 1999;4:585–595. - PubMed
-
- Beug H, von Kirchbach A, Doderlein G, Conscience JF, Graf T. Chicken hematopoietic cells transformed by seven strains of defective avian leukemia viruses display three distinct phenotypes of differentiation. Cell. 1979;18:375–390. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
