A tumor host range selection procedure identifies p150(sal2) as a target of polyoma virus large T antigen
- PMID: 11734654
- PMCID: PMC64731
- DOI: 10.1073/pnas.251447198
A tumor host range selection procedure identifies p150(sal2) as a target of polyoma virus large T antigen
Abstract
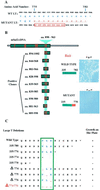
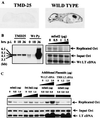
Cancer cells may undergo loss or alterations in functions that certain viruses normally target to promote virus replication. Virus mutants that have lost the targeting function(s) should be able to grow in such cancer cells but not in normal cells. A "tumor host range" (t-hr) selection procedure has been devised and applied to polyoma virus based on this rationale. Studies of one t-hr mutant have led to the identification of the mSal2 gene product (p150(sal2)) as a binding partner of the large T antigen. mSal2 encodes a multizinc finger protein and putative transcription factor homologous to the Drosophila homeotic gene Spalt. The t-hr mutant encodes an altered large T protein that fails to interact with p150(sal2) and is defective in replication and tumor induction in newborn mice.
Figures



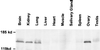
Similar articles
-
Separation of immortalization from tumor induction with polyoma large T mutants that fail to bind the retinoblastoma gene product.Oncogene. 1992 Oct;7(10):1979-87. Oncogene. 1992. PMID: 1328987
-
Lytic functions of mutant polyomavirus large T-antigen with deletion of retinoblastoma protein-binding motif.Virology. 1993 Mar;193(1):281-8. doi: 10.1006/viro.1993.1123. Virology. 1993. PMID: 8382398
-
Polyoma virus: old findings and new challenges.Virology. 2001 Oct 25;289(2):167-73. doi: 10.1006/viro.2001.1124. Virology. 2001. PMID: 11689038 Review.
-
Zinc-binding and protein-protein interactions mediated by the polyomavirus large T antigen zinc finger.J Virol. 1995 May;69(5):2842-9. doi: 10.1128/JVI.69.5.2842-2849.1995. J Virol. 1995. PMID: 7707506 Free PMC article.
-
Studies on the polyoma virus tumor-specific transplantation antigen (TSTA).Adv Cancer Res. 1990;55:57-85. doi: 10.1016/s0065-230x(08)60468-6. Adv Cancer Res. 1990. PMID: 1696775 Review. No abstract available.
Cited by
-
The tumor suppressor protein p150(Sal2) in carcinogenesis.Tumour Biol. 2015 Feb;36(2):489-94. doi: 10.1007/s13277-014-3019-1. Epub 2015 Jan 22. Tumour Biol. 2015. PMID: 25608837 Review.
-
Transcriptional and post-translational regulation of the quiescence factor and putative tumor suppressor p150(Sal2).FASEB J. 2011 Apr;25(4):1275-83. doi: 10.1096/fj.10-173674. Epub 2011 Jan 12. FASEB J. 2011. PMID: 21228219 Free PMC article.
-
Casein kinase 2 phosphorylates and induces the SALL2 tumor suppressor degradation in colon cancer cells.Cell Death Dis. 2024 Mar 16;15(3):223. doi: 10.1038/s41419-024-06591-z. Cell Death Dis. 2024. PMID: 38493149 Free PMC article.
-
The polyoma virus large T binding protein p150 is a transcriptional repressor of c-MYC.PLoS One. 2012;7(9):e46486. doi: 10.1371/journal.pone.0046486. Epub 2012 Sep 28. PLoS One. 2012. PMID: 23029531 Free PMC article.
-
Sall1, sall2, and sall4 are required for neural tube closure in mice.Am J Pathol. 2008 Nov;173(5):1455-63. doi: 10.2353/ajpath.2008.071039. Epub 2008 Sep 25. Am J Pathol. 2008. PMID: 18818376 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

