Protein kinase A regulates sexual development and gluconeogenesis through phosphorylation of the Zn finger transcriptional activator Rst2p in fission yeast
- PMID: 11739717
- PMCID: PMC134213
- DOI: 10.1128/MCB.22.1.1-11.2002
Protein kinase A regulates sexual development and gluconeogenesis through phosphorylation of the Zn finger transcriptional activator Rst2p in fission yeast
Abstract
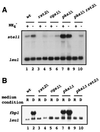
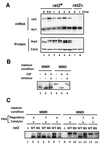
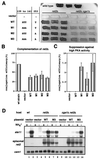
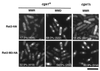
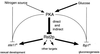
Protein kinase A (PKAi a cyclic AMP-dependent protein kinase) negatively regulates sexual development and gluconeogenesis in fission yeast by suppressing the transcription of ste11 required for the former and the transcription of fbp1 required for the latter. Here we show that Rst2p, a zinc finger protein that can bind to the upstream region of ste11 and fbp1 via the STREP motif, mediates the activity of PKA to transcription of these genes. A simple reporter system confirmed that PKA could cause its negative effect on transcription through the combination of Rst2p and STREP. Rst2p was phosphorylated by PKA in vitro at two consensus sequences on it. Substitution of the target threonine residues by alanine made the protein active even in the presence of high PKA activity. Rst2p underwent hyperphosphorylation in the medium lacking glucose, and PKA inhibited this hyperphosphorylation. Rst2p was mainly cytoplasmic under high PKA activity but was concentrated in the nucleus when this activity was lowered, suggesting that PKA might regulate ste11 and fbp1 negatively by excluding Rst2p from the nucleus. However, the shift of Rst2p localization was not perfect under physiological conditions, leaving the possibility that PKA inhibits Rst2p function in another way as well. Although the PKA-Rst2p-STREP pathway is apparently central to the regulation of ste11 and fbp1 transcription in accordance with nutritional conditions, some additional paths are likely to connect nitrogen to repression of ste11 and glucose to repression of fbp1. These paths may ensure the specificity between the type of nutrients in shortage and the type of genes to be expressed.
Figures




References
-
- Alfa, C., P. A. Fantes, J. Hyams, M. McLeod, and E. Warbrick. 1993. Experiments with fission yeast: a laboratory course manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
-
- Bähler, J., J. Q. Wu, M. S. Longtine, N. G. Shah, A. McKenzie III, A. B. Steever, A. Wach, P. Philippsen, and J. R. Pringle. 1998. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 14:943–951. - PubMed
-
- Cherry, J. R., T. R. Johnson, C. Dollard, J. R. Shuster, and C. L. Denis. 1989. Cyclic AMP-dependent protein kinase phosphorylates and inactivates the yeast transcriptional activator ADR1. Cell 56:409–419. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
