Different myrosinase and idioblast distribution in Arabidopsis and Brassica napus
- PMID: 11743118
- PMCID: PMC133578
- DOI: 10.1104/pp.010334
Different myrosinase and idioblast distribution in Arabidopsis and Brassica napus
Abstract
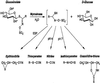
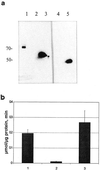
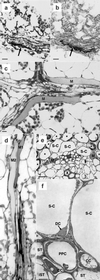
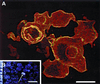
Myrosinase (EC 3.2.3.1) is a glucosinolate-degrading enzyme mainly found in special idioblasts, myrosin cells, in Brassicaceae. This two-component system of secondary products and degradative enzymes is important in plant-insect interactions. Immunocytochemical analysis of Arabidopsis localized myrosinase exclusively to myrosin cells in the phloem parenchyma, whereas no myrosin cells were detected in the ground tissue. In Brassica napus, myrosinase could be detected in myrosin cells both in the phloem parenchyma and in the ground tissue. The myrosin cells were similar in Arabidopsis and B. napus and were found to be different from the companion cells and the glucosinolate-containing S-cells present in Arabidopsis. Confocal laser scanning immunomicroscopy analysis of myrosin cells in B. napus embryos showed that the myrosin grains constitute a continuous reticular system in the cell. These findings indicate that in the two species studied, initial cells creating the ground tissue have different potential for making idioblasts and suggest that the myrosinase-glucosinolate system has at least partly different functions. Several myrosinases in B. napus extracts are recovered in complex together with myrosinase-binding protein (MBP), and the localization of MBP was therefore studied in situ. The expression of MBP was highest in germinating seedlings of B. napus and was found in every cell except the myrosin cells of the ground tissue. Rapid disappearance of the MBP from the non-myrosin cells and emergence of MBP in the myrosin cells resulted in an apparent colocalization of MBP and myrosinase in 7-d-old seedlings.
Figures



Similar articles
-
Cell specific, cross-species expression of myrosinases in Brassica napus, Arabidopsis thaliana and Nicotiana tabacum.Plant Mol Biol. 2004 Mar;54(4):597-611. doi: 10.1023/B:PLAN.0000038272.99590.10. Plant Mol Biol. 2004. PMID: 15316292
-
'Myrosin cells' are not a prerequisite for aphid feeding on oilseed rape (Brassica napus) but affect host plant preferences.Plant Biol (Stuttg). 2012 Nov;14(6):894-904. doi: 10.1111/j.1438-8677.2012.00578.x. Epub 2012 Jun 5. Plant Biol (Stuttg). 2012. PMID: 22672561
-
Removing the mustard oil bomb from seeds: transgenic ablation of myrosin cells in oilseed rape (Brassica napus) produces MINELESS seeds.J Exp Bot. 2010 Jun;61(6):1683-97. doi: 10.1093/jxb/erq039. Epub 2010 Mar 10. J Exp Bot. 2010. PMID: 20219777 Free PMC article.
-
Specialized Vacuoles of Myrosin Cells: Chemical Defense Strategy in Brassicales Plants.Plant Cell Physiol. 2018 Jul 1;59(7):1309-1316. doi: 10.1093/pcp/pcy082. Plant Cell Physiol. 2018. PMID: 29897512 Review.
-
The Cellular and Subcellular Organization of the Glucosinolate-Myrosinase System against Herbivores and Pathogens.Int J Mol Sci. 2022 Jan 29;23(3):1577. doi: 10.3390/ijms23031577. Int J Mol Sci. 2022. PMID: 35163500 Free PMC article. Review.
Cited by
-
Antifungal Effect of Brassica Tissues on the Mycotoxigenic Cereal Pathogen Fusarium graminearum.Antibiotics (Basel). 2022 Sep 15;11(9):1249. doi: 10.3390/antibiotics11091249. Antibiotics (Basel). 2022. PMID: 36140028 Free PMC article.
-
A complex interplay of three R2R3 MYB transcription factors determines the profile of aliphatic glucosinolates in Arabidopsis.Plant Physiol. 2010 May;153(1):348-63. doi: 10.1104/pp.109.149286. Epub 2010 Mar 26. Plant Physiol. 2010. PMID: 20348214 Free PMC article.
-
Management of Reniform Nematode in Cotton Using Winter Crop Residue Amendments Under Greenhouse Conditions.J Nematol. 2023 Oct 21;55(1):20230041. doi: 10.2478/jofnem-2023-0041. eCollection 2023 Feb. J Nematol. 2023. PMID: 37868787 Free PMC article.
-
Initiation of ER Body Formation and Indole Glucosinolate Metabolism by the Plastidial Retrograde Signaling Metabolite, MEcPP.Mol Plant. 2017 Nov 6;10(11):1400-1416. doi: 10.1016/j.molp.2017.09.012. Epub 2017 Sep 28. Mol Plant. 2017. PMID: 28965830 Free PMC article.
-
Indole-3-acetonitrile production from indole glucosinolates deters oviposition by Pieris rapae.Plant Physiol. 2008 Mar;146(3):916-26. doi: 10.1104/pp.107.112185. Epub 2008 Jan 11. Plant Physiol. 2008. PMID: 18192443 Free PMC article.
References
-
- Behnke HD, Eschlbeck G. Dilated cisternae in Capparales: an attempt towards the characterization of a specific endoplasmic reticulum. Protoplasma. 1978;97:351–363.
-
- Bodnaryk RP. Effects of wounding on glucosinolates in the cotyledons of oilseed rape and mustard. Phytochemistry. 1992;31:2671–2677.
-
- Bones A, Iversen TH. Myrosin cells and myrosinase. Isr J Bot. 1985;34:351–376.
-
- Bones A, Rossiter JT. The myrosinase-glucosinolate system, its organization and biochemistry. Physiol Plant. 1996;97:194–208.
-
- Bones A, Thangstad OP, Haugen OA, Espevik T. Fate of myrosin cells: characterization of monoclonal antibodies against myrosinase. J Exp Bot. 1991;42:1541–1549.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

