Endothelial transcytosis of myeloperoxidase confers specificity to vascular ECM proteins as targets of tyrosine nitration
- PMID: 11748259
- PMCID: PMC209464
- DOI: 10.1172/JCI12617
Endothelial transcytosis of myeloperoxidase confers specificity to vascular ECM proteins as targets of tyrosine nitration
Abstract
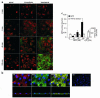
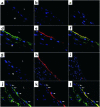
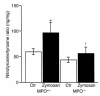
Nitrotyrosine formation is a hallmark of vascular inflammation, with polymorphonuclear neutrophil-derived (PMN-derived) and monocyte-derived myeloperoxidase (MPO) being shown to catalyze this posttranslational protein modification via oxidation of nitrite (NO(2)(-)) to nitrogen dioxide (NO(2)(*)). Herein, we show that MPO concentrates in the subendothelial matrix of vascular tissues by a transcytotic mechanism and serves as a catalyst of ECM protein tyrosine nitration. Purified MPO and MPO released by intraluminal degranulation of activated human PMNs avidly bound to aortic endothelial cell glycosaminoglycans in both cell monolayer and isolated vessel models. Cell-bound MPO rapidly transcytosed intact endothelium and colocalized abluminally with the ECM protein fibronectin. In the presence of the substrates hydrogen peroxide (H(2)O(2)) and NO(2)(-), cell and vessel wall-associated MPO catalyzed nitration of ECM protein tyrosine residues, with fibronectin identified as a major target protein. Both heparin and the low-molecular weight heparin enoxaparin significantly inhibited MPO binding and protein nitrotyrosine (NO(2)Tyr) formation in both cultured endothelial cells and rat aortic tissues. MPO(-/-) mice treated with intraperitoneal zymosan had lower hepatic NO(2)Tyr/tyrosine ratios than did zymosan-treated wild-type mice. These data indicate that MPO significantly contributes to NO(2)Tyr formation in vivo. Moreover, transcytosis of MPO, occurring independently of leukocyte emigration, confers specificity to nitration of vascular matrix proteins.
Figures


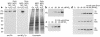

Similar articles
-
Spatial mapping of pulmonary and vascular nitrotyrosine reveals the pivotal role of myeloperoxidase as a catalyst for tyrosine nitration in inflammatory diseases.Free Radic Biol Med. 2002 Oct 1;33(7):1010. doi: 10.1016/s0891-5849(02)00993-0. Free Radic Biol Med. 2002. PMID: 12361810
-
The potentiation of myeloperoxidase activity by the glycosaminoglycan-dependent binding of myeloperoxidase to proteins of the extracellular matrix.Biochim Biophys Acta. 2013 Oct;1830(10):4524-36. doi: 10.1016/j.bbagen.2013.05.024. Epub 2013 May 23. Biochim Biophys Acta. 2013. PMID: 23707661
-
Myeloperoxidase and horseradish peroxidase catalyze tyrosine nitration in proteins from nitrite and hydrogen peroxide.Arch Biochem Biophys. 1998 Aug 15;356(2):207-13. doi: 10.1006/abbi.1998.0772. Arch Biochem Biophys. 1998. PMID: 9705211
-
Myeloperoxidase and its contributory role in inflammatory vascular disease.Pharmacol Ther. 2006 Jul;111(1):16-26. doi: 10.1016/j.pharmthera.2005.06.023. Epub 2006 Feb 13. Pharmacol Ther. 2006. PMID: 16476484 Review.
-
NO-dependent protein nitration: a cell signaling event or an oxidative inflammatory response?Trends Biochem Sci. 2003 Dec;28(12):646-54. doi: 10.1016/j.tibs.2003.10.006. Trends Biochem Sci. 2003. PMID: 14659696 Review.
Cited by
-
Low-density lipoprotein modified by myeloperoxidase in inflammatory pathways and clinical studies.Mediators Inflamm. 2013;2013:971579. doi: 10.1155/2013/971579. Epub 2013 Jul 24. Mediators Inflamm. 2013. PMID: 23983406 Free PMC article. Review.
-
Overview of Crosstalk Between Multiple Factor of Transcytosis in Blood Brain Barrier.Front Neurosci. 2020 Jan 21;13:1436. doi: 10.3389/fnins.2019.01436. eCollection 2019. Front Neurosci. 2020. PMID: 32038141 Free PMC article. Review.
-
Myeloperoxidase-derived oxidation: mechanisms of biological damage and its prevention.J Clin Biochem Nutr. 2011 Jan;48(1):8-19. doi: 10.3164/jcbn.11-006FR. Epub 2010 Dec 28. J Clin Biochem Nutr. 2011. PMID: 21297906 Free PMC article.
-
Pathogenesis of anti-neutrophil cytoplasmic antibody-associated vasculitis.Rheumatol Immunol Res. 2023 Apr 18;4(1):11-21. doi: 10.2478/rir-2023-0003. eCollection 2023 Mar. Rheumatol Immunol Res. 2023. PMID: 37138650 Free PMC article. Review.
-
Cardiac myeloperoxidase activity is elevated in hypertensive pregnant rats.J Huazhong Univ Sci Technolog Med Sci. 2017 Dec;37(6):904-909. doi: 10.1007/s11596-017-1825-6. Epub 2017 Dec 21. J Huazhong Univ Sci Technolog Med Sci. 2017. PMID: 29270751
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

