Proteasomal regulation of betac signaling reveals a novel mechanism for cytokine receptor heterotypic desensitization
- PMID: 11748263
- PMCID: PMC209471
- DOI: 10.1172/JCI13877
Proteasomal regulation of betac signaling reveals a novel mechanism for cytokine receptor heterotypic desensitization
Abstract
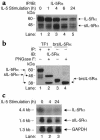
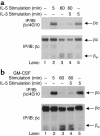
IL-5, IL-3, and GM-CSF are hematopoietic cytokines that are key mediators of the allergic inflammatory response. The receptors for these three cytokines consist of a cytokine-specific alpha (Ralpha) chain and a shared common beta (betac) chain. Herein, we demonstrate that agonistic ligation of these receptor subunits rapidly induces proteasomal degradation of the betac, but not the Ralpha, cytoplasmic domain, resulting in termination of signal transduction and yielding a truncated betac isoform ligated to the Ralpha subunit. Proteasomal degradation of the betac cytoplasmic domain was also a prerequisite for endocytosis and lysosomal degradation of the ligated receptor subunits. Moreover, proteasome-dependent termination of signaling induced by one betac-engaging cytokine resulted in cellular desensitization to signal transduction by subsequent stimulation with another betac-engaging cytokine. These data provide the first evidence for ligand-dependent proteasomal degradation of the betac cytoplasmic domain, and they establish a novel mechanism for heterotypic desensitization of shared cytokine receptor signaling.
Figures
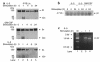




Similar articles
-
Dissection of the cytoplasmic domains of cytokine receptors involved in STAT and Ras dependent proliferation.Oncogene. 2002 May 16;21(22):3579-91. doi: 10.1038/sj.onc.1205444. Oncogene. 2002. PMID: 12032860
-
Interleukin-3-induced activation of the JAK/STAT pathway is prolonged by proteasome inhibitors.Blood. 1998 May 1;91(9):3182-92. Blood. 1998. PMID: 9558373
-
JAK kinases control IL-5 receptor ubiquitination, degradation, and internalization.J Leukoc Biol. 2007 Apr;81(4):1137-48. doi: 10.1189/jlb.0706465. Epub 2007 Jan 16. J Leukoc Biol. 2007. PMID: 17227823
-
Function and signal transduction mediated by the interleukin 3 receptor system in hematopoiesis.Stem Cells. 1996 Nov;14(6):605-18. doi: 10.1002/stem.140605. Stem Cells. 1996. PMID: 8948019 Review.
-
Signalling by the βc family of cytokines.Cytokine Growth Factor Rev. 2013 Jun;24(3):189-201. doi: 10.1016/j.cytogfr.2013.03.002. Epub 2013 Mar 25. Cytokine Growth Factor Rev. 2013. PMID: 23535386 Review.
Cited by
-
LNK/SH2B3 Loss of Function Promotes Atherosclerosis and Thrombosis.Circ Res. 2016 Sep 2;119(6):e91-e103. doi: 10.1161/CIRCRESAHA.116.308955. Epub 2016 Jul 18. Circ Res. 2016. PMID: 27430239 Free PMC article.
-
CBL linker region and RING finger mutations lead to enhanced granulocyte-macrophage colony-stimulating factor (GM-CSF) signaling via elevated levels of JAK2 and LYN.J Biol Chem. 2013 Jul 5;288(27):19459-70. doi: 10.1074/jbc.M113.475087. Epub 2013 May 21. J Biol Chem. 2013. PMID: 23696637 Free PMC article.
-
Interleukin-5 and IL-5 receptor in health and diseases.Proc Jpn Acad Ser B Phys Biol Sci. 2011;87(8):463-85. doi: 10.2183/pjab.87.463. Proc Jpn Acad Ser B Phys Biol Sci. 2011. PMID: 21986312 Free PMC article. Review.
-
Competitive regulation of E-cadherin juxtamembrane domain degradation by p120-catenin binding and Hakai-mediated ubiquitination.PLoS One. 2012;7(5):e37476. doi: 10.1371/journal.pone.0037476. Epub 2012 May 31. PLoS One. 2012. PMID: 22693575 Free PMC article.
-
Neutrophil GM-CSF receptor dynamics in acute lung injury.J Leukoc Biol. 2019 Jun;105(6):1183-1194. doi: 10.1002/JLB.3MA0918-347R. Epub 2019 Apr 3. J Leukoc Biol. 2019. PMID: 30942918 Free PMC article.
References
-
- Metcalf D. Hematopoietic regulators: redundancy or subtlety? Blood. 1993;82:3515–3523. - PubMed
-
- Arai KI, et al. Cytokines: coordinators of immune and inflammatory responses. Annu Rev Biochem. 1990;59:783–836. - PubMed
-
- Tavernier J, et al. A human high affinity interleukin-5 receptor (IL-5R) is composed of an IL-5-specific α chain and a β chain shared with the receptor for GM-CSF. Cell. 1991;66:1175–1184. - PubMed
-
- Kitamura T, Sato N, Arai K, Miyajima A. Expression cloning of the human IL-3 receptor cDNA reveals a shared β subunit for the human IL-3 and GM-CSF receptors. Cell. 1991;66:1165–1174. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

