A soluble factor(s) secreted from CD8(+) T lymphocytes inhibits human immunodeficiency virus type 1 replication through STAT1 activation
- PMID: 11752148
- PMCID: PMC136805
- DOI: 10.1128/jvi.76.2.569-581.2002
A soluble factor(s) secreted from CD8(+) T lymphocytes inhibits human immunodeficiency virus type 1 replication through STAT1 activation
Abstract
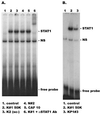
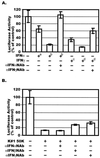
CD8(+) T lymphocytes can suppress human immunodeficiency virus type 1 (HIV-1) replication by secreting a soluble factor(s) known as CD8(+) T-lymphocyte antiviral factor (CAF). One site of CAF action is inhibition of HIV-1 RNA transcription, particularly at the step of long terminal repeat (LTR)-driven gene expression. However, the mechanism by which CAF inhibits LTR activation is not understood. Here, we show that conditioned media from several herpesvirus saimari-transformed CD8(+) T lymphocytes inhibit, in a time- and dose-dependent manner, the replication of HIV-1 pseudotype viruses that express the envelope glycoproteins of vesicular stomatitis virus (HIV-1(VSV)). The same conditioned media also inhibit phorbol myristate acetate-induced activation of the HIV-1 LTR and activate the signal transducer and activator of transcription 1 (STAT1) protein. We have obtained direct evidence that STAT1 is necessary for CAF-mediated inhibition of LTR activation and HIV-1 replication. Thus, the inhibitory effect of CAF on HIV-1(VSV) replication was abolished in STAT1-deficient cells. Moreover, CAF inhibition of LTR activation was diminished both in STAT1-deficient cells and in cells expressing a STAT1 dominant negative mutant but was restored when STAT1 was reintroduced into the STAT1-deficient cells. We also observed that CAF induced the expression of interferon regulatory factor 1 (IRF-1), and that IRF-1 gene induction was STAT-1 dependent. Taken together, our results suggest that CAF activates STAT1, leading to IRF-1 induction and inhibition of gene expression regulated by the HIV-1 LTR. This study therefore helps clarify one molecular mechanism of host defense against HIV-1.
Figures






Similar articles
-
CAF-mediated human immunodeficiency virus (HIV) type 1 transcriptional inhibition is distinct from alpha-defensin-1 HIV inhibition.J Virol. 2003 Jun;77(12):6777-84. doi: 10.1128/jvi.77.12.6777-6784.2003. J Virol. 2003. PMID: 12767998 Free PMC article.
-
Modulation of human immunodeficiency virus 1 replication by interferon regulatory factors.J Exp Med. 2002 May 20;195(10):1359-70. doi: 10.1084/jem.20010753. J Exp Med. 2002. PMID: 12021315 Free PMC article.
-
T cell-derived suppressive activity: evidence of autocrine noncytolytic control of HIV type 1 transcription and replication.AIDS Res Hum Retroviruses. 1999 Nov 20;15(17):1553-61. doi: 10.1089/088922299309847. AIDS Res Hum Retroviruses. 1999. PMID: 10580406
-
CD8+ cell-derived anti-human immunodeficiency virus inhibitory factor.J Infect Dis. 1999 May;179 Suppl 3:S485-8. doi: 10.1086/314808. J Infect Dis. 1999. PMID: 10099125 Review.
-
CD8+ T-cell-mediated non-cytolytic suppression of human immuno-deficiency viruses.Curr Drug Targets Infect Disord. 2003 Jun;3(2):97-113. doi: 10.2174/1568005033481196. Curr Drug Targets Infect Disord. 2003. PMID: 12769788 Review.
Cited by
-
Proteomic analysis of HIV-infected macrophages.J Neuroimmune Pharmacol. 2011 Mar;6(1):89-106. doi: 10.1007/s11481-010-9253-4. Epub 2010 Dec 14. J Neuroimmune Pharmacol. 2011. PMID: 21153888 Free PMC article. Review.
-
Mechanisms of CD8+ T cell-mediated suppression of HIV/SIV replication.Eur J Immunol. 2018 Jun;48(6):898-914. doi: 10.1002/eji.201747172. Epub 2018 Mar 26. Eur J Immunol. 2018. PMID: 29427516 Free PMC article. Review.
-
Macrophage derived cystatin B/cathepsin B in HIV replication and neuropathogenesis.Curr HIV Res. 2014;12(2):111-20. doi: 10.2174/1570162x12666140526120249. Curr HIV Res. 2014. PMID: 24862331 Free PMC article. Review.
-
Relative efficacy of T cell stimuli as inducers of productive HIV-1 replication in latently infected CD4 lymphocytes from patients on suppressive cART.Virology. 2017 Aug;508:127-133. doi: 10.1016/j.virol.2017.05.008. Virology. 2017. PMID: 28527342 Free PMC article.
-
Role of CXCR5+ CD8+ T cells in human immunodeficiency virus-1 infection.Front Microbiol. 2022 Nov 14;13:998058. doi: 10.3389/fmicb.2022.998058. eCollection 2022. Front Microbiol. 2022. PMID: 36452930 Free PMC article. Review.
References
-
- Bailer, R. T., A. Holloway, J. Sun, J. B. Margolick, M. Martin, J. Kostman, and L. J. Montaner. 1999. IL-13 and IFN-gamma secretion by activated T cells in HIV-1 infection associated with viral suppression and a lack of disease progression. J. Immunol. 162:7534–7542. - PubMed
-
- Benkirane, M., R. F. Chun, H. Xiao, V. V. Ogryzko, B. H. Howard, Y. Nakatani, and K. T. Jeang. 1998. Activation of integrated provirus requires histone acetyltransferase: p300 and P/CAF are coactivators for HIV-1 Tat. J. Biol. Chem. 273:24898–24905. - PubMed
-
- Bovolenta, C., L. Camorali, A. L. Lorini, S. Ghezzi, E. Vicenzi, A. Lazzarin, and G. Poli. 1999. Constitutive activation of STATs upon in vivo human immunodeficiency virus infection. Blood 94:4202–4209. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

