Evidence that the cell wall of Bacillus subtilis is protonated during respiration
- PMID: 11752466
- PMCID: PMC65017
- DOI: 10.1073/pnas.261483798
Evidence that the cell wall of Bacillus subtilis is protonated during respiration
Abstract
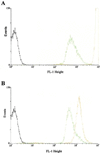
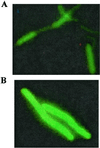
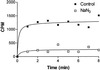
Several independent experiments suggest that cell walls of Bacillus subtilis are protonated during growth. When cells were grown in the presence of fluorescein-labeled dextran to saturate the cell walls, centrifuged, and suspended in PBS, fluorescence-activated cell sorter analyses revealed the bacteria were only poorly fluorescent. In contrast, when the bacteria were purged with N(2) to dissipate protonmotive force (pmf) fluorescence became intense. Upon reconstitution of the pmf with phenazine methosulfate, glucose, and oxygen, fluorescence declined. Another approach used pH-dependent chemical modification of cell walls. The walls of respiring B. subtilis cells were amenable to carboxylate modification by [(14)C]ethanolamine and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide. The carbodiimide activation of carboxylate groups occurs only in acidic conditions. Upon dissipation of pmf the walls were refractory to chemical modification. Ammonium groups can be condensed with FITC in alkaline medium, but the condensation is very slow in acidic buffers. It was found that the derivatization of the walls with FITC could occur in the absence of pmf. The use of pH-dependent fluorophores and pH-dependent chemical modification reactions suggest that cell walls of respiring B. subtilis cells have a relatively low pH environment. This study shows a bacterium has a protonated compartment. Acidification of cell walls during growth may be one means of regulating autolytic enzymes.
Figures

Similar articles
-
Regulation of autolysins in teichuronic acid-containing Bacillus subtilis cells.Mol Microbiol. 2002 May;44(3):601-6. doi: 10.1046/j.1365-2958.2002.02872.x. Mol Microbiol. 2002. PMID: 11994144
-
Inhibition of growth and cell wall morphogenesis of Bacillus subtilis by extracellular slime produced by Physarum flavicomum.Cytobios. 1977;20(79-80):163-77. Cytobios. 1977. PMID: 104827
-
Hydrogen ion control of autolysin-dependent functions in Bacillus subtilis.Microbios. 1983;38(153-154):187-94. Microbios. 1983. PMID: 6139741
-
Production of fungal cell wall degrading enzymes by a biocontrol strain of Bacillus subtilis AF 1.Indian J Exp Biol. 2005 Oct;43(10):892-6. Indian J Exp Biol. 2005. PMID: 16235723
-
Cell wall assembly in Bacillus subtilis: how spirals and spaces challenge paradigms.Mol Microbiol. 2006 Jun;60(5):1077-90. doi: 10.1111/j.1365-2958.2006.05169.x. Mol Microbiol. 2006. PMID: 16689786 Review.
Cited by
-
Cellular and Enzymatic Determinants Impacting the Exolytic Action of an Anti-Staphylococcal Enzybiotic.Int J Mol Sci. 2023 Dec 30;25(1):523. doi: 10.3390/ijms25010523. Int J Mol Sci. 2023. PMID: 38203699 Free PMC article.
-
Impaired respiration elicits SrrAB-dependent programmed cell lysis and biofilm formation in Staphylococcus aureus.Elife. 2017 Feb 21;6:e23845. doi: 10.7554/eLife.23845. Elife. 2017. PMID: 28221135 Free PMC article.
-
Phenazine redox cycling enhances anaerobic survival in Pseudomonas aeruginosa by facilitating generation of ATP and a proton-motive force.Mol Microbiol. 2014 Apr;92(2):399-412. doi: 10.1111/mmi.12566. Epub 2014 Mar 19. Mol Microbiol. 2014. PMID: 24612454 Free PMC article.
-
Identification of IspC, an 86-kilodalton protein target of humoral immune response to infection with Listeria monocytogenes serotype 4b, as a novel surface autolysin.J Bacteriol. 2007 Mar;189(5):2046-54. doi: 10.1128/JB.01375-06. Epub 2006 Dec 15. J Bacteriol. 2007. PMID: 17172332 Free PMC article.
-
A continuum of anionic charge: structures and functions of D-alanyl-teichoic acids in gram-positive bacteria.Microbiol Mol Biol Rev. 2003 Dec;67(4):686-723. doi: 10.1128/MMBR.67.4.686-723.2003. Microbiol Mol Biol Rev. 2003. PMID: 14665680 Free PMC article. Review.
References
-
- Koch A L. J Theor Biol. 1986;120:73–84. - PubMed
-
- Shockman G D, Holtje J-V. In: Bacterial Cell Wall. Ghuysen J-M, Hakenbeck R, editors. Amsterdam: Elsevier; 1994. pp. 131–166.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

