Brca2 (XRCC11) deficiency results in radioresistant DNA synthesis and a higher frequency of spontaneous deletions
- PMID: 11756561
- PMCID: PMC139737
- DOI: 10.1128/MCB.22.2.669-679.2002
Brca2 (XRCC11) deficiency results in radioresistant DNA synthesis and a higher frequency of spontaneous deletions
Abstract
We show here that the radiosensitive Chinese hamster cell mutant (V-C8) of group XRCC11 is defective in the breast cancer susceptibility gene Brca2. The very complex phenotype of V-C8 cells is complemented by a single human chromosome 13 providing the BRCA2 gene, as well as by the murine Brca2 gene. The Brca2 deficiency in V-C8 cells causes hypersensitivity to various DNA-damaging agents with an extreme sensitivity toward interstrand DNA cross-linking agents. Furthermore, V-C8 cells show radioresistant DNA synthesis after ionizing radiation, suggesting that Brca2 deficiency affects cell cycle checkpoint regulation. In addition, V-C8 cells display tremendous chromosomal instability and a high frequency of abnormal centrosomes. The mutation spectrum at the hprt locus showed that the majority of spontaneous mutations in V-C8 cells are deletions, in contrast to wild-type V79 cells. A mechanistic explanation for the genome instability phenotype of Brca2-deficient cells is provided by the observation that the nuclear localization of the central DNA repair protein in homologous recombination, Rad51, is reduced in V-C8 cells.
Figures
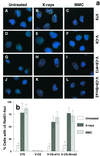



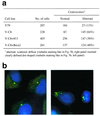

References
-
- Bhattacharyya, A., U. S. Ear, B. H. Koller, R. R. Weichselbaum, and D. K. Bishop. 2000. The breast cancer susceptibility gene BRCA1 is required for subnuclear assembly of Rad51 and survival following treatment with the DNA cross-linking agent cisplatin. J. Biol. Chem. 275:23899–23903. - PubMed
-
- Bignell, G., G. Micklem, M. R. Stratton, A. Ashworth, and R. Wooster. 1997. The BRC repeats are conserved in mammalian BRCA2 proteins. Hum. Mol. Genet. 6:53–58. - PubMed
-
- Bishop, D. K., U. Ear, A. Bhattacharyya, C. Calderone, M. Beckett, R. R. Weichselbaum, and A. Shinohara. 1998. Xrcc3 is required for assembly of Rad51 complexes in vivo. J. Biol. Chem. 273:21482–21488. - PubMed
-
- Davies, A. A., J. Y. Masson, M. J. McIlwraith, A. Z. Stasiak, A. Stasiak, A. R. Venkitaraman, and S. C. West. 2001. Role of BRCA2 in control of the RAD51 recombination and DNA repair protein. Mol. Cell 7:273–282. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
