Soluble fusion proteins between single transmembrane photoreceptor guanylyl cyclases and their activators
- PMID: 11772023
- PMCID: PMC1363675
- DOI: 10.1021/bi015606u
Soluble fusion proteins between single transmembrane photoreceptor guanylyl cyclases and their activators
Abstract
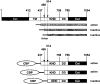
Among single-spanning transmembrane receptors (sTMRs), two guanylyl cyclase receptors, GC1 and GC2, are critically important during phototransduction in vertebrate retinal photoreceptor cells. Ca(2+)-free forms of guanylyl cyclase-activating proteins (GCAPs) stimulate GCs intracellularly by a molecular mechanism that is not fully understood. To gain further insight into the mechanism of activation and specificity among these proteins, for the first time, several soluble and active truncated GCs and fusion proteins between intracellular domains of GCs and full-length GCAPs were generated. The GC activity of myristoylated GCAP--(437-1054)GC displayed typical [Ca(2+)] dependence, and was further enhanced by ATP and inhibited by guanylyl cyclase inhibitor protein (GCIP). The myristoyl group of GCAP1 appeared to be critical for the inhibition of GCs at high [Ca(2+)], even without membranes. In contrast, calmodulin (CaM)--(437-1054)GC1 fusion protein was inactive, but could be stimulated by exogenous GCAP1. In a series of experiments, we showed that the activation of GCs by linked GCAPs involved intra- and intermolecular mechanisms. The catalytically productive GCAP1--(437-1054)GC1 complex can dissociate, allowing binding and stimulation of the GC1 fusion protein by free GCAP1. This suggests that the intramolecular interactions within the fusion protein have low affinity and are mimicking the native system. We present evidence that the mechanism of GC activation by GCAPs involves a dimeric form of GCs, involves direct interaction between GCs and GCAPs, and does not require membrane components. Thus, fusion proteins may provide an important advance for further structural studies of photoreceptor GCs and other sTMRs with and without different forms of regulatory proteins.
Figures


Similar articles
-
Guanylyl cyclase activating protein. A calcium-sensitive regulator of phototransduction.J Biol Chem. 1995 Sep 15;270(37):22029-36. doi: 10.1074/jbc.270.37.22029. J Biol Chem. 1995. PMID: 7665624
-
Identification of functional regions of guanylate cyclase-activating protein 1 (GCAP1) using GCAP1/GCIP chimeras.Biol Chem. 2001 Aug;382(8):1179-88. doi: 10.1515/BC.2001.148. Biol Chem. 2001. PMID: 11592399
-
Impairment of the rod outer segment membrane guanylate cyclase dimerization in a cone-rod dystrophy results in defective calcium signaling.Biochemistry. 2000 Oct 17;39(41):12522-33. doi: 10.1021/bi001514d. Biochemistry. 2000. PMID: 11027131
-
Regulation of photoreceptor membrane guanylyl cyclases by guanylyl cyclase activator proteins.Methods. 1999 Dec;19(4):521-31. doi: 10.1006/meth.1999.0894. Methods. 1999. PMID: 10581151 Review.
-
Photoreceptor specific guanylate cyclases in vertebrate phototransduction.Mol Cell Biochem. 2002 Jan;230(1-2):97-106. Mol Cell Biochem. 2002. PMID: 11952100 Review.
Cited by
-
A novel mutation (I143NT) in guanylate cyclase-activating protein 1 (GCAP1) associated with autosomal dominant cone degeneration.Invest Ophthalmol Vis Sci. 2004 Nov;45(11):3863-70. doi: 10.1167/iovs.04-0590. Invest Ophthalmol Vis Sci. 2004. PMID: 15505030 Free PMC article.
-
Photoreceptor guanylate cyclase variants: cGMP production under control.Acta Biochim Pol. 2003;50(4):1075-95. Acta Biochim Pol. 2003. PMID: 14739996 Free PMC article. Review.
-
A novel GCAP1 missense mutation (L151F) in a large family with autosomal dominant cone-rod dystrophy (adCORD).Invest Ophthalmol Vis Sci. 2005 Apr;46(4):1124-32. doi: 10.1167/iovs.04-1431. Invest Ophthalmol Vis Sci. 2005. PMID: 15790869 Free PMC article.
-
Ca2+ -dependent regulation of phototransduction.Photochem Photobiol. 2008 Jul-Aug;84(4):903-10. doi: 10.1111/j.1751-1097.2008.00323.x. Epub 2008 Mar 12. Photochem Photobiol. 2008. PMID: 18346093 Free PMC article. Review.
References
-
- Pawson T, Gish GD. Cell. 1992;71:359–362. - PubMed
-
- Polans A, Baehr W, Palczewski K. Trends Neurosci. 1996;19:547–554. - PubMed
-
- Palczewski K, Polans AS, Baehr W, Ames JB. Bioessays. 2000;22:337–350. - PubMed
-
- Lucas KA, Pitari GM, Kazerounian S, Ruiz-Stewart I, Park J, Schulz S, Chepenik KP, Waldman SA. Pharmacol. Rev. 2000;52:375–414. - PubMed
-
- Kelsell RE, Gregory Evans K, Payne AM, Perrault I, Kaplan J, Yang RB. Hum. Mol. Genet. 1998;7:1179–1184. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

