Developmental aspects of human hepatic drug glucuronidation in young children and adults
- PMID: 11788570
- PMCID: PMC1773113
- DOI: 10.1136/gut.50.2.259
Developmental aspects of human hepatic drug glucuronidation in young children and adults
Abstract
Background and aims: The liver represents one of the major sites of human glucuronidation. Many therapeutic drugs are substrates for UDP-glucuronosyltransferases (UGT) leading to the formation of usually inactive glucuronides. Hepatic glucuronidation undergoes significant changes during fetal and neonatal development requiring age adapted drug therapy. Regulation of individual UGT genes during hepatic development has not been defined.
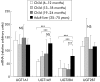
Subjects and methods: Expression of 13 UGT genes and glucuronidation activities were analysed in 16 paediatric liver samples (aged 7-24 months), two fetal samples, and 12 adult liver samples (aged 25-75 years) using duplex reverse transcription-polymerase chain reaction, western blot, and specific catalytic UGT activity assays.
Results: No UGT transcripts were detected in fetal liver at 20 weeks' gestation. In contrast, UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B4, UGT2B7, UGT2B10, and UGT2B15 transcripts were present without variation in all 28 hepatic samples after six months of age. Significantly lower expression of UGT1A9 and UGT2B4 mRNA was identified in paediatric liver. Hepatic glucuronidation activity in children aged 13-24 months was found to be lower than in adults for ibuprofen (24-fold), amitriptyline (16-fold), 4-tert-butylphenol (40-fold), estrone (15-fold), and buprenorphine (12-fold).
Conclusions: An early phase characterised by the appearance of UGT gene transcripts and a later phase characterised by upregulation of UGT expression is demonstrated during human hepatic development. The differential regulation of UGT1A9 and UGT2B4 expression extends beyond two years of age and is capable of influencing hepatic glucuronidation of common therapeutic drugs in children. The development of hepatic UGT activities is significant for paediatric drug therapy and the prevention of adverse drug effects.
Figures



References
-
- Dutton GJ. Glucuronidation of drugs and other compounds. Boca Raton, Florida: CRC Boca Raton Press, 1980.
-
- Tukey RH, Strassburg CP. Human UDP-glucuronosyltransferases: metabolism, expression, and disease. Annu Rev Pharmacol Toxicol 2000;40:581–616. - PubMed
-
- Burchell B, Nebert DW, Nelson DR, et al. The UDP glucuronosyltransferase gene superfamily: suggested nomenclature based on evolutionary divergence. DNA Cell Biol 1991;10:487–94. - PubMed
-
- Strassburg CP, Manns MP, Tukey RH. Differential down-regulation of the UDP-glucuronosyltransferase 1A locus is an early event in human liver and biliary cancer. Cancer Res 1997;57:2979–85. - PubMed
-
- Strassburg CP, Oldhafer K, Manns MP, et al. Differential expression of the UGT1A locus in human liver, biliary, and gastric tissue: identification of UGT1A7 and UGT1A10 transcripts in extrahepatic tissue. Mol Pharmacol 1997;52:212–20. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
