TEA- and apamin-resistant K(Ca) channels in guinea-pig myenteric neurons: slow AHP channels
- PMID: 11790810
- PMCID: PMC2290069
- DOI: 10.1113/jphysiol.2001.012952
TEA- and apamin-resistant K(Ca) channels in guinea-pig myenteric neurons: slow AHP channels
Abstract
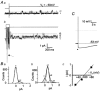
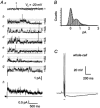
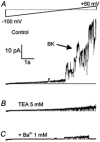
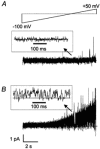
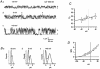
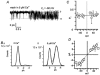
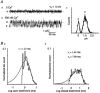
The patch-clamp technique was used to record from intact ganglia of the guinea-pig duodenum in order to characterize the K(+) channels that underlie the slow afterhyperpolarization (slow AHP) of myenteric neurons. Cell-attached patch recordings from slow AHP-generating (AH) neurons revealed an increased open probability (P(o)) of TEA-resistant K(+) channels following action potentials. The P(o) increased from < 0.06 before action potentials to 0.33 in the 2 s following action potential firing. The ensemble averaged current had a similar time course to the current underlying the slow AHP. TEA- and apamin-resistant Ca(2+)-activated K(+) (K(Ca)) channels were present in inside-out patches excised from AH neurons. The P(o) of these channels increased from < 0.03 to approximately 0.5 upon increasing cytoplasmic [Ca(2+)] from < 10 nM to either 500 nM or 10 microM. P(o) was insensitive to changes in transpatch potential. The unitary conductance of these TEA- and apamin-resistant K(Ca) channels measured approximately 60 pS under symmetric K(+) concentrations between -60 mV and +60 mV, but decreased outside this range. Under asymmetrical [K(+)], the open channel current showed outward rectification and had a limiting slope conductance of about 40 pS. Activation of the TEA- and apamin-resistant K(Ca) channels by internal Ca(2+) in excised patches was not reversed by washing out the Ca(2+)-containing solution and replacing it with nominally Ca(2+)-free physiological solution. Kinetic analysis of the single channel recordings of the TEA- and apamin-resistant K(Ca) channels was consistent with their having a single open state of about 2 ms (open dwell time distribution was fitted with a single exponential) and at least two closed states (two exponential functions were required to adequately fit the closed dwell time distribution). The Ca(2+) dependence of the activation of TEA- and apamin-resistant K(Ca) channels resides in the long-lived closed state which decreased from > 100 ms in the absence of Ca(2+) to about 7 ms in the presence of submicromolar cytoplasmic Ca(2+). The Ca(2+)-insensitive closed dwell time had a time constant of about 1 ms. We propose that these small-to-intermediate conductance TEA- and apamin-resistant Ca(2+)-activated K(+) channels are the channels that are primarily responsible for the slow AHP in myenteric AH neurons.
Figures


References
-
- Bekkers JM. Distribution of slow AHP channels on hippocampal CA1 pyramidal neurons. Journal of Neurophysiology. 2000;83:1756–1759. - PubMed
-
- Bertrand PP, Kunze WA A, Bornstein JC, Furness JB. Electrical mapping of the projections of intrinsic primary afferent neurons to the mucosa of the guinea-pig small intestine. Neurogastroenterology. 1998;10:533–541. - PubMed
-
- Bond CT, Maylie J, Adelman JP. Small-conductance calcium-activated potassium channels. Annals of the New York Academy of Sciences. 1999;868:370–378. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

