Intracellular survival of Leishmania major in neutrophil granulocytes after uptake in the absence of heat-labile serum factors
- PMID: 11796617
- PMCID: PMC127667
- DOI: 10.1128/IAI.70.2.826-835.2002
Intracellular survival of Leishmania major in neutrophil granulocytes after uptake in the absence of heat-labile serum factors
Abstract
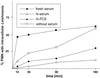
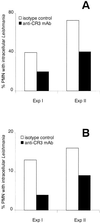
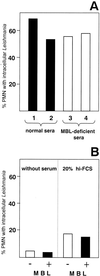
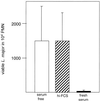
The role of polymorphonuclear neutrophil granulocytes (PMN) in defense against the intracellular parasite Leishmania is poorly understood. In the present study, the interaction of human PMN with Leishmania major promastigotes was investigated in vitro. In the presence of fresh human serum, about 50% of PMN phagocytosed the parasites within 10 min and the parasite uptake led to PMN activation, resulting in the killing of most ingested parasites. Heat inactivation of the serum markedly reduced the rate of early parasite phagocytosis, suggesting a role of complement components in the early uptake of Leishmania. However, over 50% of PMN were able to ingest parasites in the presence of heat-inactivated serum if the coincubation was extended to 3 h. After 3 h, 10% of the PMN were found to internalize Leishmania even under serum-free conditions. These findings indicate that PMN possess mechanisms for both opsonin/complement-dependent and -independent uptake of Leishmania. Both pathways of uptake could be partially blocked by anti-CR3 antibody. Mannan-binding lectin was found not to be involved in this process. When phagocytosed in the absence of opsonin, the majority of Leishmania parasites survived intracellularly in PMN for at least 1 day. These data suggest a dual role of PMN in the early response to L. major infection. On the one hand, PMN can rapidly eliminate the intracellular parasites, and on the other hand, Leishmania can survive intracellularly in PMN. These data, together with the finding that intact parasites were seen in PMN isolated from the skin of infected mice, suggest that PMN can serve as host cells for the intracellular survival of Leishmania within the first hours or days after infection.
Figures



Similar articles
-
Inhibition of the spontaneous apoptosis of neutrophil granulocytes by the intracellular parasite Leishmania major.J Immunol. 2002 Jul 15;169(2):898-905. doi: 10.4049/jimmunol.169.2.898. J Immunol. 2002. PMID: 12097394
-
Cutting edge: neutrophil granulocyte serves as a vector for Leishmania entry into macrophages.J Immunol. 2004 Dec 1;173(11):6521-5. doi: 10.4049/jimmunol.173.11.6521. J Immunol. 2004. PMID: 15557140
-
Leishmania promastigotes release a granulocyte chemotactic factor and induce interleukin-8 release but inhibit gamma interferon-inducible protein 10 production by neutrophil granulocytes.Infect Immun. 2002 Aug;70(8):4177-84. doi: 10.1128/IAI.70.8.4177-4184.2002. Infect Immun. 2002. PMID: 12117926 Free PMC article.
-
Neutrophil granulocytes as host cells and transport vehicles for intracellular pathogens: apoptosis as infection-promoting factor.Immunobiology. 2008;213(3-4):183-91. doi: 10.1016/j.imbio.2007.11.010. Epub 2008 Feb 8. Immunobiology. 2008. PMID: 18406366 Review.
-
Apoptosis driven infection.Autoimmunity. 2007 Jun;40(4):349-52. doi: 10.1080/08916930701356960. Autoimmunity. 2007. PMID: 17516227 Review.
Cited by
-
Twin Attributes of Tyrosyl-tRNA Synthetase of Leishmania donovani: A HOUSEKEEPING PROTEIN TRANSLATION ENZYME AND A MIMIC OF HOST CHEMOKINE.J Biol Chem. 2016 Aug 19;291(34):17754-71. doi: 10.1074/jbc.M116.727107. Epub 2016 Jul 5. J Biol Chem. 2016. PMID: 27382051 Free PMC article.
-
Complement C3 is required for the progression of cutaneous lesions and neutrophil attraction in Leishmania major infection.Med Microbiol Immunol. 2005 May;194(3):143-9. doi: 10.1007/s00430-004-0229-y. Epub 2004 Sep 17. Med Microbiol Immunol. 2005. PMID: 15378355
-
Leishmania amazonensis promastigotes induce and are killed by neutrophil extracellular traps.Proc Natl Acad Sci U S A. 2009 Apr 21;106(16):6748-53. doi: 10.1073/pnas.0900226106. Epub 2009 Apr 3. Proc Natl Acad Sci U S A. 2009. PMID: 19346483 Free PMC article.
-
Ex Vivo Analysis of the Association of GFP-Expressing L. aethiopica and L. mexicana with Human Peripheral Blood-Derived (PBD) Leukocytes over 24 Hours.Microorganisms. 2024 Sep 19;12(9):1909. doi: 10.3390/microorganisms12091909. Microorganisms. 2024. PMID: 39338584 Free PMC article.
-
The state of art of neutrophil extracellular traps in protozoan and helminthic infections.Biosci Rep. 2019 Jan 11;39(1):BSR20180916. doi: 10.1042/BSR20180916. Print 2019 Jan 31. Biosci Rep. 2019. PMID: 30498092 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

