Rapid activation of protein tyrosine kinase and phospholipase C-gamma2 and increase in cytosolic free calcium are required by Ehrlichia chaffeensis for internalization and growth in THP-1 cells
- PMID: 11796624
- PMCID: PMC127685
- DOI: 10.1128/IAI.70.2.889-898.2002
Rapid activation of protein tyrosine kinase and phospholipase C-gamma2 and increase in cytosolic free calcium are required by Ehrlichia chaffeensis for internalization and growth in THP-1 cells
Abstract
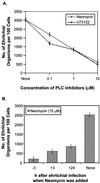
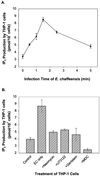
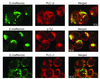
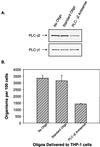
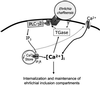
Ehrlichia chaffeensis, a bacterium that cannot survive outside the eukaryotic cell, proliferates exclusively in human monocytes and macrophages. In this study, signaling events required for ehrlichial infection of human monocytic cell line THP-1 were characterized. Entry and proliferation of E. chaffeensis in THP-1 cells were significantly blocked by various inhibitors that can regulate calcium signaling, including 8-(diethylamino)octyl-3,4,5-trimethoxybenzoate and 2-aminoethoxydiphenyl borate (intracellular calcium mobilization inhibitors), verapamil and 1-[beta-[3-(4-methoxyphenyl)propyl]-4-methoxyphenethyl]-1H-imidazole (SKF-96365) (calcium channel inhibitors), neomycin and 1-(6-[[17beta-3-methoxyestra-1,3,5(10)-trien-17-yl]amino]hexyl)-1H-pyrrole-2,5-dione (U-73122) (phospholipase C [PLC] inhibitors), monodansylcadaverine (a transglutaminase [TGase] inhibitor), and genistein (a protein tyrosine kinase [PTK] inhibitor). Addition of E. chaffeensis resulted in rapid increases in the level of inositol 1,4,5-trisphosphate (IP(3)) and the level of cytosolic free calcium ([Ca(2+)](i)) in THP-1 cells, which were prevented by pretreatment of THP-1 cells with inhibitors of TGase, PTK, and PLC. E. chaffeensis induced rapid tyrosine phosphorylation of PLC-gamma2, and the presence of a PLC-gamma2 antisense oligonucleotide in THP-1 cells significantly blocked ehrlichial infection. Furthermore, tyrosine-phosphorylated proteins and PLC-gamma2 were colocalized with ehrlichial inclusions, as determined by double-immunofluorescence labeling. The heat-sensitive component of viable E. chaffeensis cells was essential for these signaling events. E. chaffeensis, therefore, can recruit interacting signal-transducing molecules and induce the following signaling events required for the establishment of infection in host cells: protein cross-linking by TGase, tyrosine phosphorylation, PLC-gamma2 activation, IP(3) production, and an increase in [Ca(2+)](i).
Figures

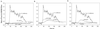

Similar articles
-
Protein kinase A-mediated inhibition of gamma interferon-induced tyrosine phosphorylation of Janus kinases and latent cytoplasmic transcription factors in human monocytes by Ehrlichia chaffeensis.Infect Immun. 1998 Jun;66(6):2514-20. doi: 10.1128/IAI.66.6.2514-2520.1998. Infect Immun. 1998. PMID: 9596710 Free PMC article.
-
Calcium-sensing receptor-mediated activation of phospholipase C-gamma1 is downstream of phospholipase C-beta and protein kinase C in MC3T3-E1 osteoblasts.Bone. 2002 Apr;30(4):559-66. doi: 10.1016/s8756-3282(01)00700-1. Bone. 2002. PMID: 11934646
-
Mechanism of vascular smooth muscle cells activation by hydrogen peroxide: role of phospholipase C gamma.Nephrol Dial Transplant. 2002 Mar;17(3):392-8. doi: 10.1093/ndt/17.3.392. Nephrol Dial Transplant. 2002. PMID: 11865083
-
Regulation of the phospholipase C-gamma2 pathway in B cells.Immunol Rev. 2000 Aug;176:19-29. doi: 10.1034/j.1600-065x.2000.00605.x. Immunol Rev. 2000. PMID: 11043765 Review.
-
Beyond calcium: new signaling pathways for Tec family kinases.J Cell Sci. 2002 Aug 1;115(Pt 15):3039-48. doi: 10.1242/jcs.115.15.3039. J Cell Sci. 2002. PMID: 12118060 Review.
Cited by
-
Type IV secretion in the obligatory intracellular bacterium Anaplasma phagocytophilum.Cell Microbiol. 2010 Sep 1;12(9):1213-21. doi: 10.1111/j.1462-5822.2010.01500.x. Epub 2010 Jul 28. Cell Microbiol. 2010. PMID: 20670295 Free PMC article. Review.
-
Bacterial Manipulation of Wnt Signaling: A Host-Pathogen Tug-of-Wnt.Front Immunol. 2019 Oct 17;10:2390. doi: 10.3389/fimmu.2019.02390. eCollection 2019. Front Immunol. 2019. PMID: 31681283 Free PMC article. Review.
-
Global proteomic analysis of two tick-borne emerging zoonotic agents: anaplasma phagocytophilum and ehrlichia chaffeensis.Front Microbiol. 2011 Feb 17;2:24. doi: 10.3389/fmicb.2011.00024. eCollection 2011. Front Microbiol. 2011. PMID: 21687416 Free PMC article.
-
Restoring Light Sensitivity in Blind Retinae Using a Photochromic AMPA Receptor Agonist.ACS Chem Neurosci. 2016 Jan 20;7(1):15-20. doi: 10.1021/acschemneuro.5b00234. Epub 2015 Nov 3. ACS Chem Neurosci. 2016. PMID: 26495755 Free PMC article.
-
Ehrlichia chaffeensis Exploits Canonical and Noncanonical Host Wnt Signaling Pathways To Stimulate Phagocytosis and Promote Intracellular Survival.Infect Immun. 2015 Dec 28;84(3):686-700. doi: 10.1128/IAI.01289-15. Infect Immun. 2015. PMID: 26712203 Free PMC article.
References
-
- Abe, S., K. Yamashita, H. Kohno, and Y. Ohkubo. 2000. Involvement of transglutaminase in the receptor-mediated endocytosis of mouse peritoneal macrophages. Biol. Pharm. Bull. 23:1511–1513. - PubMed
-
- Andersson, S. G., A. Zomorodipour, J. O. Andersson, T. Sicheritz-Ponten, U. C. Alsmark, R. M. Podowski, A. K. Naslund, A. S. Eriksson, H. H. Winkler, and C. G. Kurland. 1998. The genome sequence of Rickettsia prowazekii and the origin of mitochondria. Nature 396:133–140. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

