VP40, the matrix protein of Marburg virus, is associated with membranes of the late endosomal compartment
- PMID: 11799178
- PMCID: PMC135914
- DOI: 10.1128/jvi.76.4.1825-1838.2002
VP40, the matrix protein of Marburg virus, is associated with membranes of the late endosomal compartment
Abstract
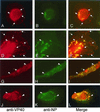
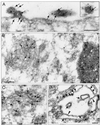
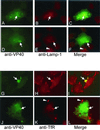
Localization of VP40 in Marburg virus (MBGV)-infected cells was studied by using immunofluorescence and immunoelectron microscopic analysis. VP40 was detected in association with nucleocapsid structures, present in viral inclusions and at sites of virus budding. Additionally, VP40 was identified in the foci of virus-induced membrane proliferation and in intracellular membrane clusters which had the appearance of multivesicular bodies (MVBs). VP40-containing MVBs were free of nucleocapsids. When analyzed by immunogold labeling, the concentration of VP40 in MVBs was six times higher than in nucleocapsid structures. Biochemical studies showed that recombinant VP40 represented a peripheral membrane protein that was stably associated with membranes by hydrophobic interaction. Recombinant VP40 was also found in association with membranes of MVBs and in filopodia- or lamellipodia-like protrusions at the cell surface. Antibodies against marker proteins of various cellular compartments showed that VP40-positive membranes contained Lamp-1 and the transferrin receptor, confirming that they belong to the late endosomal compartment. VP40-positive membranes were also associated with actin. Western blot analysis of purified MBGV structural proteins demonstrated trace amounts of actin, Lamp-1, and Rab11 (markers of recycling endosomes), while markers for other cellular compartments were absent. Our data indicate that MBGV VP40 was able to interact with membranes of late endosomes in the course of viral infection. This capability was independent of other MBGV proteins.
Figures



References
-
- Becker, S., H.-D. Klenk, and E. Mühlberger. 1996. Intracellular transport and processing of the Marburg virus surface protein in vertebrate and insect cells. Virology 225:145-155. - PubMed
-
- Becker, S., C. Rinne, U. Hofsäss, H.-D. Klenk, and E. Mühlberger. 1998. Interactions of Marburg virus nucleocapsid proteins. Virology 249:406-417. - PubMed
-
- Bordier, C. 1981. Phase separation of integral membrane proteins in Triton X-114 solution. J. Biol. Chem. 256:1604-1607. - PubMed
-
- Bretscher, M. S., and C. Aguado-Velasco. 1998. Membrane traffic during cell locomotion. Curr. Opin. Cell Biol. 10:537-541. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

