RelB nuclear translocation mediated by C-terminal activator regions of Epstein-Barr virus-encoded latent membrane protein 1 and its effect on antigen-presenting function in B cells
- PMID: 11799186
- PMCID: PMC135895
- DOI: 10.1128/jvi.76.4.1914-1921.2002
RelB nuclear translocation mediated by C-terminal activator regions of Epstein-Barr virus-encoded latent membrane protein 1 and its effect on antigen-presenting function in B cells
Abstract
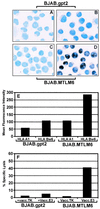
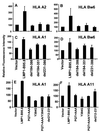
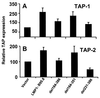
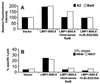
Previous studies have shown that Epstein-Barr virus-encoded latent membrane protein 1 (LMP1) is uniquely able to up-regulate the expression of the peptide transporters (referred to as TAP-1 and TAP-2) and major histocompatibility complex (MHC) class I in Burkitt's lymphoma (BL) cell lines. This up-regulation is often accompanied by a restoration of antigen-presenting function as measured by the ability of these cells to present endogenously expressed viral antigen to cytotoxic T lymphocytes. Here we show that the expression of LMP1 resulted in up-regulation and nuclear translocation of RelB that were coincident with increased expression of MHC class I in BL cells. Deletion of the C-terminal activator regions (CTARs) of LMP1 significantly impaired the abilities of LMP1 to translocate RelB into the nucleus and to up-regulate the expression of antigen-processing genes. Further analysis with single-point mutations within the CTARs confirmed that the residues critical for NF-kappa B activation directly contribute to antigen-processing function regulation in BL cells. This LMP1-mediated effect was blocked following expression of either dominant negative I kappa B alpha S32/36A, an NF-kappa B inhibitor, or antisense RelB. These observations indicate that upregulation of antigen-presenting function in B cells mediated by LMP1 is signaled through the NF-kappa B subunit RelB. The data provide a mechanism by which LMP1 modulates immunogenicity of Epstein-Barr virus-infected normal and malignant cells.
Figures

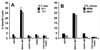
Similar articles
-
Latent membrane protein 1 inhibits Epstein-Barr virus lytic cycle induction and progress via different mechanisms.J Virol. 2003 Apr;77(8):5000-7. doi: 10.1128/jvi.77.8.5000-5007.2003. J Virol. 2003. PMID: 12663807 Free PMC article.
-
Restoration of endogenous antigen processing in Burkitt's lymphoma cells by Epstein-Barr virus latent membrane protein-1: coordinate up-regulation of peptide transporters and HLA-class I antigen expression.Eur J Immunol. 1995 May;25(5):1374-84. doi: 10.1002/eji.1830250536. Eur J Immunol. 1995. PMID: 7774641
-
Epstein-Barr virus latent membrane protein-1 activates CD25 expression in lymphoma cells involving the NFkappaB pathway.Genes Immun. 2001 Dec;2(8):433-41. doi: 10.1038/sj.gene.6363803. Genes Immun. 2001. PMID: 11781710
-
[Molecular biological properties of the Epstein-Barr virus LMP1 gene: structure, function and polymorphism].Vopr Virusol. 2015;60(3):5-13. Vopr Virusol. 2015. PMID: 26281300 Review. Russian.
-
Role of LMP1 in immune control of EBV infection.Semin Cancer Biol. 2001 Dec;11(6):455-60. doi: 10.1006/scbi.2001.0412. Semin Cancer Biol. 2001. PMID: 11669607 Review.
Cited by
-
The Possible Future Roles for iPSC-Derived Therapy for Autoimmune Diseases.J Clin Med. 2015 May 28;4(6):1193-206. doi: 10.3390/jcm4061193. J Clin Med. 2015. PMID: 26239553 Free PMC article. Review.
-
Latent membrane protein 1 inhibits Epstein-Barr virus lytic cycle induction and progress via different mechanisms.J Virol. 2003 Apr;77(8):5000-7. doi: 10.1128/jvi.77.8.5000-5007.2003. J Virol. 2003. PMID: 12663807 Free PMC article.
-
Epstein-Barr virus deubiquitinase downregulates TRAF6-mediated NF-κB signaling during productive replication.J Virol. 2013 Apr;87(7):4060-70. doi: 10.1128/JVI.02020-12. Epub 2013 Jan 30. J Virol. 2013. PMID: 23365429 Free PMC article.
-
Dendritic cells and the promise of antigen-specific therapy in rheumatoid arthritis.Arthritis Res Ther. 2013 Feb 4;15(1):204. doi: 10.1186/ar4130. Arthritis Res Ther. 2013. PMID: 23374912 Free PMC article. Review.
-
Analysis of NF-κB Pathway Proteins in Pediatric Hodgkin Lymphoma: Correlations with EBV Status and Clinical Outcome-A Children's Oncology Group Study.Lymphoma. 2012;2012:341629. doi: 10.1155/2012/341629. Lymphoma. 2012. PMID: 31406604 Free PMC article.
References
-
- Brodeur, S. R., G. Cheng, D. Baltimore, and D. A. Thorley-Lawson. 1997. Localization of the major NF-κB-activating site and the sole TRAF3 binding site of LMP-1 defines two distinct signaling motifs. J. Biol. Chem. 272:19777-19784. - PubMed
-
- Burrows, S. R., J. Gardner, R. Khanna, T. Steward, D. J. Moss, S. Rodda, and A. Suhrbier. 1994. Five new cytotoxic T cell epitopes identified within Epstein-Barr virus nuclear antigen 3. J. Gen. Virol. 75:2489-2493. - PubMed
-
- Burrows, S. R., S. J. Rodda, A. Suhrbier, H. M. Geysen, and D. J. Moss. 1992. The specificity of recognition of a cytotoxic T lymphocyte epitope. Eur. J. Immunol. 22:191-195. - PubMed
-
- Cherney, B. W., C. Sgadari, C. Kanegane, F. Wang, and G. Tosato. 1998. Expression of the Epstein-Barr virus protein LMP1 mediates tumor regression in vivo. Blood 91:2491-2500. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

