Evidence for coordinated interaction of cyclin D3 with p21 and cdk6 in directing the development of uterine stromal cell decidualization and polyploidy during implantation
- PMID: 11804782
- PMCID: PMC4322898
- DOI: 10.1016/s0925-4773(01)00614-1
Evidence for coordinated interaction of cyclin D3 with p21 and cdk6 in directing the development of uterine stromal cell decidualization and polyploidy during implantation
Abstract
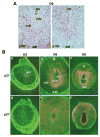
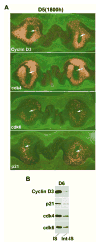
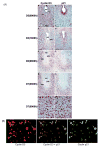
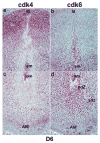
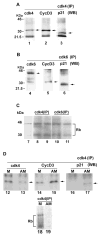
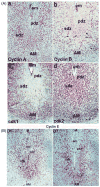
Uterine decidualization, characterized by stromal cell proliferation, and differentiation into specialized type of cells (decidual cells) with polyploidy, during implantation is critical to the pregnancy establishment in mice. The mechanisms by which the cell cycle events govern these processes are poorly understood. The cell cycle is tightly regulated at two particular checkpoints, G1-S and G2-M phases. Normal operation of these phases involves a complex interplay of cyclins, cyclin-dependent kinases (cdks) and cdk inhibitors (CKIs). We previously observed that upregulation of uterine cyclin D3 at the implantation site is tightly associated with decidualization in mice. To better understand the role of cyclin D3 in this process, we examined cell-specific expression and associated interactions of several cell cycle regulators (cyclins, cdks and CKIs) specific to different phases of the cell cycle during decidualization in mice. Among the various cell cycle molecules examined, coordinate expression and functional association of cyclin D3 with cdk4 suggest a role for proliferation and, that of cyclin D3 with p21 and cdk6 is consistent with the development of polyploidy during stromal cell decidualization.
Figures


Similar articles
-
HB-EGF directs stromal cell polyploidy and decidualization via cyclin D3 during implantation.Dev Biol. 2004 Jan 1;265(1):181-95. doi: 10.1016/j.ydbio.2003.09.019. Dev Biol. 2004. PMID: 14697362 Free PMC article.
-
Induction of cell cycle regulatory proteins in anti-immunoglobulin-stimulated mature B lymphocytes.J Exp Med. 1996 Aug 1;184(2):407-17. doi: 10.1084/jem.184.2.407. J Exp Med. 1996. PMID: 8760794 Free PMC article.
-
Control of regional decidualization in implantation: Role of FoxM1 downstream of Hoxa10 and cyclin D3.Sci Rep. 2015 Sep 9;5:13863. doi: 10.1038/srep13863. Sci Rep. 2015. PMID: 26350477 Free PMC article.
-
Cell cycle regulatory control for uterine stromal cell decidualization in implantation.Reproduction. 2009 Jun;137(6):889-99. doi: 10.1530/REP-08-0539. Epub 2009 Mar 23. Reproduction. 2009. PMID: 19307426 Review.
-
Cyclins and CDKS in development and cancer: lessons from genetically modified mice.Front Biosci. 2006 Jan 1;11:1164-88. doi: 10.2741/1871. Front Biosci. 2006. PMID: 16146805 Review.
Cited by
-
The decidua-the maternal bed embracing the embryo-maintains the pregnancy.Semin Immunopathol. 2016 Nov;38(6):635-649. doi: 10.1007/s00281-016-0574-0. Epub 2016 Jun 10. Semin Immunopathol. 2016. PMID: 27287066 Free PMC article. Review.
-
The Multiple Functions of HB-EGF in Female Reproduction and Related Cancer: Molecular Mechanisms and Targeting Strategies.Reprod Sci. 2024 Sep;31(9):2588-2603. doi: 10.1007/s43032-024-01454-6. Epub 2024 Feb 29. Reprod Sci. 2024. PMID: 38424408 Review.
-
Single-Cell RNA-Sequencing Reveals Interactions between Endometrial Stromal Cells, Epithelial Cells, and Lymphocytes during Mouse Embryo Implantation.Int J Mol Sci. 2022 Dec 22;24(1):213. doi: 10.3390/ijms24010213. Int J Mol Sci. 2022. PMID: 36613656 Free PMC article.
-
Homeobox genes for embryo implantation: From mouse to human.Animal Model Exp Med. 2018 Apr 19;1(1):14-22. doi: 10.1002/ame2.12002. eCollection 2018 Mar. Animal Model Exp Med. 2018. PMID: 30891542 Free PMC article. Review.
-
Spatial transcriptomic profiles of mouse uterine microenvironments at pregnancy day 7.5†.Biol Reprod. 2022 Aug 9;107(2):529-545. doi: 10.1093/biolre/ioac061. Biol Reprod. 2022. PMID: 35357464 Free PMC article.
References
-
- Ansell JD, Barlow PW, McLaren A. Binucleate and polyploid cells in the decidua of the mouse. J Embryol Experi Morphol. 1974;31:223–227. - PubMed
-
- Bamberger AM, Sudahl S, Bamberger CM, Schulte HM, Lo ning T. Expression patterns of the cell-cycle inhibitor p27 and the cell cycle promoter cyclin E in the human placenta throughout gestation: Implications for the control of proliferation. Placenta. 1999;20:401–406. - PubMed
-
- Das SK, Lim H, Paria BC, Dey SK. Cyclin D3 in the mouse uterus is associated with the decidualization process during early pregnancy. J Mol Endocrinol. 1999;22:91–101. - PubMed
-
- Deng C, Zhang P, Harper JW, Elledge SJ, Lader P. Mice lacking p21Cip1/Waf1 undergo normal development, but are defective in G1 checkpoint control. Cell. 1995;82:675–684. - PubMed
-
- Dey SK. Implantation. In: Adashi EY, Rock JA, Rosenwaks Z, editors. Reproductive Endocrinology, Surgery and Technology. Lippincott-Raven Publishers; New York: 1996. pp. 421–434.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

