Leptin modulates the effects of acyl CoA:diacylglycerol acyltransferase deficiency on murine fur and sebaceous glands
- PMID: 11805129
- PMCID: PMC150839
- DOI: 10.1172/JCI13880
Leptin modulates the effects of acyl CoA:diacylglycerol acyltransferase deficiency on murine fur and sebaceous glands
Abstract
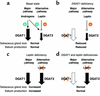
Acyl CoA:diacylglycerol acyltransferase (DGAT) is a ubiquitously expressed enzyme that catalyzes the final reaction in the major pathways of triglyceride synthesis. Mice lacking DGAT1 (Dgat(-/-)) demonstrate significant changes in lipid metabolism in several tissues, including the skin. Here we report the effects of DGAT1 deficiency on fur and sebaceous glands. Adult Dgat(-/-) mice had dry fur and hair loss, which were associated with atrophic sebaceous glands and fur lipid abnormalities. As a result, Dgat(-/-) mice had impaired water repulsion and defective thermoregulation after water immersion. These phenotypes were mostly absent in Dgat(-/-) mice with leptin deficiency, indicating an unexpected role for leptin in modulating the skin phenotype. Our findings indicate that DGAT1 plays an important role in normal fur and sebaceous gland physiology and provide evidence that leptin modulates these processes in the skin.
Figures







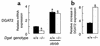
Similar articles
-
Increased insulin and leptin sensitivity in mice lacking acyl CoA:diacylglycerol acyltransferase 1.J Clin Invest. 2002 Apr;109(8):1049-55. doi: 10.1172/JCI14672. J Clin Invest. 2002. PMID: 11956242 Free PMC article.
-
Pharmacological inhibition of DGAT1 induces sebaceous gland atrophy in mouse and dog skin while overt alopecia is restricted to the mouse.Toxicol Pathol. 2015 Apr;43(3):376-83. doi: 10.1177/0192623314545657. Epub 2014 Aug 11. Toxicol Pathol. 2015. PMID: 25112279
-
Deficiency of acyl coenzyme a:diacylglycerol acyltransferase 1 increases leptin sensitivity in murine obesity models.Endocrinology. 2002 Aug;143(8):2893-8. doi: 10.1210/endo.143.8.8941. Endocrinology. 2002. PMID: 12130553
-
Inhibition of triglyceride synthesis as a treatment strategy for obesity: lessons from DGAT1-deficient mice.Arterioscler Thromb Vasc Biol. 2005 Mar;25(3):482-6. doi: 10.1161/01.ATV.0000151874.81059.ad. Epub 2004 Nov 29. Arterioscler Thromb Vasc Biol. 2005. PMID: 15569818 Review.
-
The role of acyl-CoA:diacylglycerol acyltransferase (DGAT) in energy metabolism.Ann Med. 2004;36(4):252-61. doi: 10.1080/07853890410028429. Ann Med. 2004. PMID: 15224651 Review.
Cited by
-
Loss of the acyl-CoA binding protein (Acbp) results in fatty acid metabolism abnormalities in mouse hair and skin.J Invest Dermatol. 2007 Jan;127(1):16-23. doi: 10.1038/sj.jid.5700511. Epub 2006 Aug 10. J Invest Dermatol. 2007. PMID: 16902415 Free PMC article.
-
Emerging Role of Dermal White Adipose Tissue in Modulating Hair Follicle Development During Aging.Front Cell Dev Biol. 2021 Oct 15;9:728188. doi: 10.3389/fcell.2021.728188. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34722509 Free PMC article.
-
Unique mode of lipogenic activation in rat preputial sebocytes.J Nutr Metab. 2011;2011:163631. doi: 10.1155/2011/163631. Epub 2011 Jul 26. J Nutr Metab. 2011. PMID: 21808727 Free PMC article.
-
Genetic control of de novo lipogenesis: role in diet-induced obesity.Crit Rev Biochem Mol Biol. 2010 Jun;45(3):199-214. doi: 10.3109/10409231003667500. Crit Rev Biochem Mol Biol. 2010. PMID: 20218765 Free PMC article. Review.
-
Identification of novel tissue-specific genes by analysis of microarray databases: a human and mouse model.PLoS One. 2013 May 31;8(5):e64483. doi: 10.1371/journal.pone.0064483. Print 2013. PLoS One. 2013. PMID: 23741331 Free PMC article.
References
-
- Bell RM, Coleman RA. Enzymes of glycerolipid synthesis in eukaryotes. Annu Rev Biochem. 1980;49:459–487. - PubMed
-
- Lehner R, Kuksis A. Biosynthesis of triacylglycerols. Prog Lipid Res. 1996;35:169–201. - PubMed
-
- Cases S, et al. Cloning of DGAT2, a second mammalian diacylglycerol acyltransferase, and related family members. J Biol Chem. 2001;276:38870–38876. - PubMed
-
- Farese RV, Jr, Cases S, Smith SJ. Triglyceride synthesis: insights from the cloning of diacylglycerol acyltransferase. Curr Opin Lipidol. 2000;11:229–234. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

