Protein folding mediated by solvation: water expulsion and formation of the hydrophobic core occur after the structural collapse
- PMID: 11805324
- PMCID: PMC117366
- DOI: 10.1073/pnas.022387699
Protein folding mediated by solvation: water expulsion and formation of the hydrophobic core occur after the structural collapse
Abstract
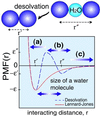
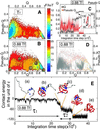
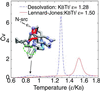
The interplay between structure-search of the native structure and desolvation in protein folding has been explored using a minimalist model. These results support a folding mechanism where most of the structural formation of the protein is achieved before water is expelled from the hydrophobic core. This view integrates water expulsion effects into the funnel energy landscape theory of protein folding. Comparisons to experimental results are shown for the SH3 protein. After the folding transition, a near-native intermediate with partially solvated hydrophobic core is found. This transition is followed by a final step that cooperatively squeezes out water molecules from the partially hydrated protein core.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

