Protein kinase Cepsilon actin-binding site is important for neurite outgrowth during neuronal differentiation
- PMID: 11809819
- PMCID: PMC65069
- DOI: 10.1091/mbc.01-04-0210
Protein kinase Cepsilon actin-binding site is important for neurite outgrowth during neuronal differentiation
Abstract
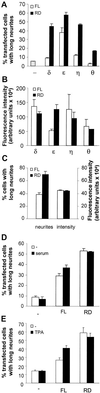
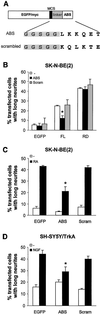
We have previously shown that protein kinase Cepsilon (PKCepsilon) induces neurite outgrowth via its regulatory domain and independently of its kinase activity. This study aimed at identifying mechanisms regulating PKCepsilon-mediated neurite induction. We show an increased association of PKCepsilon to the cytoskeleton during neuronal differentiation. Furthermore, neurite induction by overexpression of full-length PKCepsilon is suppressed if serum is removed from the cultures or if an actin-binding site is deleted from the protein. A peptide corresponding to the PKCepsilon actin-binding site suppresses neurite outgrowth during neuronal differentiation and outgrowth elicited by PKCepsilon overexpression. Neither serum removal, deletion of the actin-binding site, nor introduction of the peptide affects neurite induction by the isolated regulatory domain. Membrane targeting by myristoylation renders full-length PKCepsilon independent of both serum and the actin-binding site, and PKCepsilon colocalized with F-actin at the cortical cytoskeleton during neurite outgrowth. These results demonstrate that the actin-binding site is of importance for signals acting on PKCepsilon in a pathway leading to neurite outgrowth. Localization of PKCepsilon to the plasma membrane and/or the cortical cytoskeleton is conceivably important for its effect on neurite outgrowth.
Figures





Similar articles
-
PKCepsilon, via its regulatory domain and independently of its catalytic domain, induces neurite-like processes in neuroblastoma cells.J Cell Biol. 1999 May 17;145(4):713-26. doi: 10.1083/jcb.145.4.713. J Cell Biol. 1999. PMID: 10330401 Free PMC article.
-
Induction of neurites by the regulatory domains of PKCdelta and epsilon is counteracted by PKC catalytic activity and by the RhoA pathway.Exp Cell Res. 2004 Jan 1;292(1):135-50. doi: 10.1016/j.yexcr.2003.08.013. Exp Cell Res. 2004. PMID: 14720513
-
Ceramide influences neurite outgrowth and neuroblastoma cell apoptosis regulated by novel protein kinase C isoforms.J Neurochem. 2004 Jun;89(6):1427-35. doi: 10.1111/j.1471-4159.2004.02431.x. J Neurochem. 2004. PMID: 15189345
-
Protein kinase C and the regulation of the actin cytoskeleton.Cell Signal. 2006 Mar;18(3):276-84. doi: 10.1016/j.cellsig.2005.07.010. Epub 2005 Aug 16. Cell Signal. 2006. PMID: 16109477 Review.
-
Phospholipase D signalling and its involvement in neurite outgrowth.Biochim Biophys Acta. 2009 Sep;1791(9):898-904. doi: 10.1016/j.bbalip.2009.03.010. Epub 2009 Mar 31. Biochim Biophys Acta. 2009. PMID: 19341813 Review.
Cited by
-
Direct binding of RalA to PKCη and its crucial role in morphological change during keratinocyte differentiation.Mol Biol Cell. 2011 Apr 15;22(8):1340-52. doi: 10.1091/mbc.E10-09-0754. Epub 2011 Feb 23. Mol Biol Cell. 2011. PMID: 21346190 Free PMC article.
-
From Drosophila development to adult: clues to Notch function in long-term memory.Front Cell Neurosci. 2013 Nov 21;7:222. doi: 10.3389/fncel.2013.00222. eCollection 2013. Front Cell Neurosci. 2013. PMID: 24312012 Free PMC article.
-
Impact of PKCε downregulation on autophagy in glioblastoma cells.BMC Cancer. 2018 Feb 13;18(1):185. doi: 10.1186/s12885-018-4095-1. BMC Cancer. 2018. PMID: 29439667 Free PMC article.
-
PRKCE gene encoding protein kinase C-epsilon-Dual roles at sarcomeres and mitochondria in cardiomyocytes.Gene. 2016 Sep 15;590(1):90-6. doi: 10.1016/j.gene.2016.06.016. Epub 2016 Jun 13. Gene. 2016. PMID: 27312950 Free PMC article. Review.
-
A novel PIP2 binding of epsilonPKC and its contribution to the neurite induction ability.J Neurochem. 2007 Sep;102(5):1635-1644. doi: 10.1111/j.1471-4159.2007.04702.x. J Neurochem. 2007. PMID: 17697049 Free PMC article.
References
-
- Ács P, Bögi K, Lorenzo PS, Marquez AM, Bíró T, Szállási Z, Blumberg PM. The catalytic domain of protein kinase C chimeras modulates the affinity and targeting of phorbol ester-induced translocation. J Biol Chem. 1997a;272:22148–22153. - PubMed
-
- Ács P, Wang QJ, Bögi K, Marquez AM, Lorenzo PS, Bíró T, Szállási Z, Mushinski JF, Blumberg PM. Both the catalytic and regulatory domains of protein kinase C chimeras modulate the proliferative properties of NIH 3T3 cells. J Biol Chem. 1997b;272:28793–28799. - PubMed
-
- Aroca P, Santos E, Kazanietz MG. Recombinant C1b domain of PKCδ triggers meiotic maturation upon microinjection in Xenopus laevisoocytes. FEBS Lett. 2000;483:27–32. - PubMed
-
- Blobe GC, Stribling DS, Fabbro D, Stabel S, Hannun YA. Protein kinase C βII specifically binds to and is activated by F-actin. J Biol Chem. 1996;271:15823–15830. - PubMed
-
- Brodie C, Bogi K, Ács P, Lazarovici P, Petrovics G, Anderson WB, Blumberg PM. Protein kinase C-ε plays a role in neurite outgrowth in response to epidermal growth factor and nerve growth factor in PC12 cells. Cell Growth Differ. 1999;10:183–191. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

