A specific protein substrate for a deubiquitinating enzyme: Liquid facets is the substrate of Fat facets
- PMID: 11825870
- PMCID: PMC155328
- DOI: 10.1101/gad.961502
A specific protein substrate for a deubiquitinating enzyme: Liquid facets is the substrate of Fat facets
Abstract
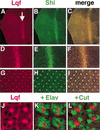
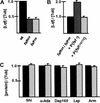
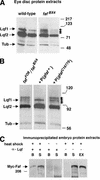
Eukaryotic genomes encode large families of deubiquitinating enzymes (DUBs). Genetic data suggest that Fat facets (Faf), a Drosophila DUB essential for patterning the compound eye, might have a novel regulatory function; Faf might reverse the ubiquitination of a specific substrate, thereby preventing proteasomal degradation of that protein. Additional genetic data implicate Liquid facets (Lqf), a homolog of the vertebrate endocytic protein epsin, as a candidate for the key substrate of Faf. Here, biochemical experiments critical to testing this model were performed. The results show definitively that Lqf is the key substrate of Faf in the eye; Lqf concentration is Faf-dependent, Lqf is ubiquitinated in vivo and deubiquitinated by Faf, and Lqf and Faf interact physically.
Figures

References
-
- Baker RT, Tobias JW, Varshavsky A. Ubiquitin-specific proteases of Saccharomyces cerevisiae: Cloning of UBP2 and UBP3, and functional analysis of the UBP gene family. J Biol Chem. 1992;267:23364–23375. - PubMed
-
- Cadavid ALM. “Identification, cloning and characterization of the Drosophila liquid facets gene.” Ph.D. thesis. Austin, TX: The University of Texas at Austin; 2000.
-
- Cadavid ALM, Ginzel A, Fischer JA. The function of the Drosophila Fat facets deubiquitinating enzyme in limiting photoreceptor cell number is intimately associated with endocytosis. Development. 2000;127:1727–1736. - PubMed
-
- Cagan RL, Kramer H, Hart AC, Zipursky SL. The Bride-of-sevenless and Sevenless interaction: Internalization of a transmembrane ligand. Cell. 1992;69:393–399. - PubMed
-
- Chen H, Fre S, Slepnev VI, Capua MR, Takei K, Butler MH, Di Fiore PP, De Camilli P. Epsin is an EH-domain-binding protein implicated in clathrin-mediated endocytosis. Nature. 1998;394:793–797. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
