Partial ablations of the flocculus and ventral paraflocculus in monkeys cause linked deficits in smooth pursuit eye movements and adaptive modification of the VOR
- PMID: 11826056
- PMCID: PMC2629758
- DOI: 10.1152/jn.00768.2000
Partial ablations of the flocculus and ventral paraflocculus in monkeys cause linked deficits in smooth pursuit eye movements and adaptive modification of the VOR
Abstract
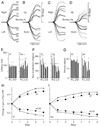
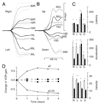
The vestibuloocular reflex (VOR) generates compensatory eye movements to stabilize visual images on the retina during head movements. The amplitude of the reflex is calibrated continuously throughout life and undergoes adaptation, also called motor learning, when head movements are persistently associated with image motion. Although the floccular-complex of the cerebellum is necessary for VOR adaptation, it is not known whether this function is localized in its anterior or posterior portions, which comprise the ventral paraflocculus and flocculus, respectively. The present paper reports the effects of partial lesions of the floccular-complex in five macaque monkeys, made either surgically or with stereotaxic injection of 3-nitropropionic acid (3-NP). Before and after the lesions, smooth pursuit eye movements were tested during sinusoidal and step-ramp target motion. Cancellation of the VOR was tested by moving a target exactly with the monkey during sinusoidal head rotation. The control VOR was tested during sinusoidal head rotation in the dark and during 30 degrees/s pulses of head velocity. VOR adaptation was studied by having the monkeys wear x2 or x0.25 optics for 4-7 days. In two monkeys, bilateral lesions removed all of the flocculus except for parts of folia 1 and 2 but did not produce any deficits in smooth pursuit, VOR adaptation, or VOR cancellation. We conclude that the flocculus alone probably is not necessary for either pursuit or VOR learning. In two monkeys, unilateral lesions including a large fraction of the ventral paraflocculus produced small deficits in horizontal and vertical smooth pursuit, and mild impairments of VOR adaptation and VOR cancellation. We conclude that the ventral paraflocculus contributes to both behaviors. In one monkey, a bilateral lesion of the flocculus and ventral paraflocculus produced severe deficits smooth pursuit and VOR cancellation, and a complete loss of VOR adaptation. Considering all five cases together, there was a strong correlation between the size of the deficits in VOR learning and pursuit. We found the strongest correlation between the behavior deficits and the size of the lesion of the ventral paraflocculus, a weaker but significant correlation for the full floccular complex, and no correlation with the size of the lesion of the flocculus. We conclude that 1) lesions of the floccular complex cause linked deficits in smooth pursuit and VOR adaptation, and 2) the relevant portions of the structure are primarily in the ventral paraflocculus, although the flocculus may participate.
Figures





Similar articles
-
Role of the cerebellar flocculus region in cancellation of the VOR during passive whole body rotation.J Neurophysiol. 2000 Sep;84(3):1599-613. doi: 10.1152/jn.2000.84.3.1599. J Neurophysiol. 2000. PMID: 10980030
-
Responses during eye movements of brain stem neurons that receive monosynaptic inhibition from the flocculus and ventral paraflocculus in monkeys.J Neurophysiol. 1994 Aug;72(2):909-27. doi: 10.1152/jn.1994.72.2.909. J Neurophysiol. 1994. PMID: 7983546
-
Neural basis for motor learning in the vestibuloocular reflex of primates. II. Changes in the responses of horizontal gaze velocity Purkinje cells in the cerebellar flocculus and ventral paraflocculus.J Neurophysiol. 1994 Aug;72(2):954-73. doi: 10.1152/jn.1994.72.2.954. J Neurophysiol. 1994. PMID: 7983548
-
Computational study on monkey VOR adaptation and smooth pursuit based on the parallel control-pathway theory.J Neurophysiol. 2002 Apr;87(4):2176-89. doi: 10.1152/jn.00168.2001. J Neurophysiol. 2002. PMID: 11929935 Review.
-
Cerebellar Control of Eye Movements.J Neuroophthalmol. 2017 Mar;37(1):87-98. doi: 10.1097/WNO.0000000000000456. J Neuroophthalmol. 2017. PMID: 27643747 Review.
Cited by
-
Specific vermal complex spike responses build up during the course of smooth-pursuit adaptation, paralleling the decrease of performance error.Exp Brain Res. 2010 Aug;205(1):41-55. doi: 10.1007/s00221-010-2331-2. Epub 2010 Jun 24. Exp Brain Res. 2010. PMID: 20574686
-
The vestibulo-ocular reflex as a model system for motor learning: what is the role of the cerebellum?Cerebellum. 2004;3(3):188-92. doi: 10.1080/14734220410018120. Cerebellum. 2004. PMID: 15543809 Review.
-
Cerebellum and ocular motor control.Front Neurol. 2011 Sep 1;2:53. doi: 10.3389/fneur.2011.00053. eCollection 2011. Front Neurol. 2011. PMID: 21909334 Free PMC article.
-
Cerebellar cortex granular layer interneurons in the macaque monkey are functionally driven by mossy fiber pathways through net excitation or inhibition.PLoS One. 2013 Dec 20;8(12):e82239. doi: 10.1371/journal.pone.0082239. eCollection 2013. PLoS One. 2013. PMID: 24376524 Free PMC article.
-
Participation of the caudal cerebellar lobule IX to the dorsal attentional network.Cerebellum Ataxias. 2018 Jun 15;5:9. doi: 10.1186/s40673-018-0088-8. eCollection 2018. Cerebellum Ataxias. 2018. PMID: 29942533 Free PMC article.
References
-
- Büttner U, Waespe W. Purkinje cell activity in the primate flocculus during optokinetic stimulation, smooth pursuit eye movements, and VOR-suppression. Exp Brain Res. 1984;55:97–104. - PubMed
-
- Carl JR, Gellman RS. Human smooth pursuit: stimulus dependent responses. J Neurophysiol. 1987;57:1446–1463. - PubMed
-
- Chyi T, Chang C. Temporal evolution of 3-nitropropionic acid-induced neurodegeneration in the rat brain by T2-weighted, diffusion-weighted, and perfusion magnetic resonance imaging. Neuroscience. 1999;92:1035–1041. - PubMed
-
- de Zeeuw CI, Hansel C, Bian F, Koekkoek SK, van Alphen AM, Linden DJ, Oberdick J. Expression of a protein kinase C inhibitor in Purkinje cells blocks cerebellar LTD and adaptation of the vestibulo-ocular reflex. Neuron. 1998;20:495–508. - PubMed
-
- Fuchs AF, Kimm J. Unit activity in the vestibular nucleus of the alert monkey during horizontal angular acceleration and eye movement. J Neurophysiol. 1975;38:1140–1161. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

