Remodelling inactivation gating of Kv4 channels by KChIP1, a small-molecular-weight calcium-binding protein
- PMID: 11826158
- PMCID: PMC2290090
- DOI: 10.1113/jphysiol.2001.013127
Remodelling inactivation gating of Kv4 channels by KChIP1, a small-molecular-weight calcium-binding protein
Abstract
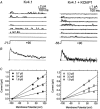
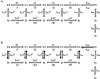
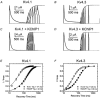
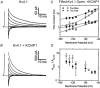
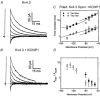
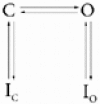
Calcium-binding proteins dubbed KChIPs favour surface expression and modulate inactivation gating of neuronal and cardiac A-type Kv4 channels. To investigate their mechanism of action, Kv4.1 or Kv4.3 were expressed in Xenopus laevis oocytes, either alone or together with KChIP1, and the K+ currents were recorded using the whole-oocyte voltage-clamp and patch-clamp methods. KChIP1 similarly remodels gating of both channels. At positive voltages, KChIP1 slows the early phase of the development of macroscopic inactivation. By contrast, the late phase is accelerated, which allows complete inactivation in < 500 ms. Thus, superimposed traces from control and KChIP1-remodelled currents crossover. KChIP1 also accelerates closed-state inactivation and recovery from inactivation (3- to 5-fold change). The latter effect is dominating and, consequently, the prepulse inactivation curves exhibit depolarizing shifts (DeltaV = 4-12 mV). More favourable closed-state inactivation may also contribute to the overall faster inactivation at positive voltages because Kv4 channels significantly inactivate from the preopen closed state. KChIP1 favours this pathway further by accelerating channel closing. The peak G-V curves are modestly leftward shifted in the presence of KChIP1, but the apparent 'threshold' voltage of current activation remains unaltered. Single Kv4.1 channels exhibited multiple conductance levels that ranged between 1.8 and 5.6 pS in the absence of KChIP1 and between 1.9 and 5.3 pS in its presence. Thus, changes in unitary conductance do not contribute to current upregulation by KChIP1. An allosteric kinetic model explains the kinetic changes by assuming that KChIP1 mainly impairs open-state inactivation, favours channel closing and lowers the energy barrier of closed-state inactivation.
Figures


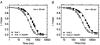



References
-
- An WF, Bowlby MR, Betty M, Cao J, Ling HP, Mendoza G, Hinson JW, Mattsson KI, Strassle BW, Trimmer JS, Rhodes KJ. Modulation of A-type potassium channels by a family of calcium sensors. Nature. 2000;403:553–556. - PubMed
-
- Bähring R, Dannenberg J, Peters HC, Leicher T, Pongs O, Isbrandt D. Conserved Kv4 N-terminal domain critical for effects of Kv channel-interacting protein 2. 2 on channel expression and gating. Journal of Biological Chemistry. 2001b;29:23888–23894. - PubMed
-
- Beck EJ, Bowlby MR, An WF, Rhodes KJ, Covarrubias M. Modulation of Kv4 inactivation gating by a calcium binding protein KChIP-1. Biophysical Journal. 2001;80:439A.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

