Pirlindole and dehydropirlindole protect rat cultured neuronal cells against oxidative stress-induced cell death through a mechanism unrelated to MAO-A inhibition
- PMID: 11834619
- PMCID: PMC1573183
- DOI: 10.1038/sj.bjp.0704519
Pirlindole and dehydropirlindole protect rat cultured neuronal cells against oxidative stress-induced cell death through a mechanism unrelated to MAO-A inhibition
Abstract
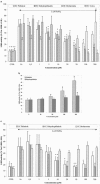
It has been shown that the MAO (monoamine oxidase)-B inhibitor deprenyl (DPR, selegiline) protects some cell types against oxidative stress. By decreasing H(2)O(2) production, MAO-A inhibitors could also reduce oxidative stress. This study reports the effect of the MAO-A inhibitors, pirlindole (PIR), dehydropirlindole (DHP), brofaromine (BRO) and moclobemide (MCL) on primary-cultured brain cells exposed to iron-mediated toxicity. A comparison with trolox (TRO), a hydrosoluble vitamin-E analogue that protects against such an induced stress, was performed. Rat hippocampal or cortical cultured cells were exposed either to 2 microM FeSO(4) alone or in the presence of PIR, DHP, BRO, DPR, MCL or TRO. Cell survival (lactate-dehydrogenase measurements, 16 h incubation), intracellular peroxide production (DCF-fluorescence, 1 h incubation), lipoperoxidation (TBARS-fluorescence, 6 h incubation) and mitochondrial function (MTT-test, 16 h incubation) were assessed. PIR, DHP and TRO significantly protected cultures (P<0.05) against Fe(2+)-induced toxicity in a concentration-dependent manner. The EC(50s) of these compounds were 6, 12 and 19 microM, respectively, in hippocampal cells. For cortical cell cultures incubated in the presence of iron and PIR or DHP, EC(50s) were 5 and 6 microM respectively. All Hill coefficients were close to unity. BRO, MCL and DPR were not protective in any type of culture. The IC(50s) for the inhibition of MAO-A were 2, 2 and 0.2 microM for PIR, DHP and BRO, respectively. PIR, DHP and TRO, but not DPR, induced a significant decrease in both intracellular peroxide production and lipoperoxidation. They also improved mitochondrial function. These experiments show that PIR and DHP can protect hippocampal and cortical neurons against oxidative stress at pharmacologically relevant concentrations. This protective effect seems unrelated to inhibition of MAO-A, but possibly involves free radical scavenging.
Figures


Similar articles
-
Pre- and post-treatment with pirlindole and dehydropirlindole protects cultured brain cells against nitric oxide-induced death.Eur J Pharmacol. 2003 Apr 11;466(1-2):21-30. doi: 10.1016/s0014-2999(03)01539-5. Eur J Pharmacol. 2003. PMID: 12679138
-
Effect of MAO-B inhibition against ischemia-induced oxidative stress in the rat brain. Comparison with a rational antioxidant.Arzneimittelforschung. 2008;58(4):160-7. doi: 10.1055/s-0031-1296487. Arzneimittelforschung. 2008. PMID: 18540477
-
Comparative effects of dehydropirlindole and other compounds on rat brain monoamine oxidase type A.Prog Neuropsychopharmacol Biol Psychiatry. 2002 Jan;26(1):75-9. doi: 10.1016/s0278-5846(01)00232-9. Prog Neuropsychopharmacol Biol Psychiatry. 2002. PMID: 11853123
-
Bifunctional drug derivatives of MAO-B inhibitor rasagiline and iron chelator VK-28 as a more effective approach to treatment of brain ageing and ageing neurodegenerative diseases.Mech Ageing Dev. 2005 Feb;126(2):317-26. doi: 10.1016/j.mad.2004.08.023. Mech Ageing Dev. 2005. PMID: 15621213 Review.
-
(-)-Deprenyl, a selective MAO-B inhibitor, with apoptotic and anti-apoptotic properties.Neurotoxicology. 2004 Jan;25(1-2):233-42. doi: 10.1016/S0161-813X(03)00102-5. Neurotoxicology. 2004. PMID: 14697898 Review.
Cited by
-
Knocking down of the KCC2 in rat hippocampal neurons increases intracellular chloride concentration and compromises neuronal survival.J Physiol. 2011 May 15;589(Pt 10):2475-96. doi: 10.1113/jphysiol.2010.203703. Epub 2011 Mar 21. J Physiol. 2011. PMID: 21486764 Free PMC article.
-
Race and Sex Differences in the Incidence and Prognostic Significance of Silent Myocardial Infarction in the Atherosclerosis Risk in Communities (ARIC) Study.Circulation. 2016 May 31;133(22):2141-8. doi: 10.1161/CIRCULATIONAHA.115.021177. Epub 2016 May 16. Circulation. 2016. PMID: 27185168 Free PMC article.
-
Moclobemide exerts anti-inflammatory effect in lipopolysaccharide-activated primary mixed glial cell culture.Naunyn Schmiedebergs Arch Pharmacol. 2010 Dec;382(5-6):409-17. doi: 10.1007/s00210-010-0535-4. Epub 2010 Sep 2. Naunyn Schmiedebergs Arch Pharmacol. 2010. PMID: 20811738
-
Structure Prediction and Potential Inhibitors Docking of Enterovirus 2C Proteins.Front Microbiol. 2022 Apr 29;13:856574. doi: 10.3389/fmicb.2022.856574. eCollection 2022. Front Microbiol. 2022. PMID: 35572704 Free PMC article.
References
-
- ANDERSON M.C., WALDMEIMER P.C., TIPTON K.F. The inhibition of monoamine oxidase by Brofaromine. Biochem. Pharmacol. 1991;41:1871–1877. - PubMed
-
- BLANC E.M., KELLY J.F., MARK R.J., WAEG G., MATTSON M.P. 4-hydroxynonenal, an aldehydic product of lipid peroxidation, impairs signal transduction associated with the muscarinic acetylcholine and metabotropic glutamate receptors: possible action on Gαq-11. J. Neurochem. 1997;69:570–580. - PubMed
-
- BOLAND A., DELAPIERRE D., MOSSAY D., HANS P., DRESSE A. Propofol protects cultured brain cells from iron-induced death: comparison with trolox. Eur. J. Pharmacol. 2000;404:21–27. - PubMed
-
- BRUHWYLER J., LIÉGEOIS J.F., GECZY J. Pirlindole: a selective reversible inhibitor of monoamine oxidase A. A review of its preclinical properties. Pharmacol. Res. 1997;36:23–33. - PubMed
-
- BRUHWYLER J., LIÉGEOIS J.F., GÉRARDY J., DAMAS J., CHLEIDE E., LEJEUNE C., DECAMP E., DE TULLIO P., DELARGE J., DRESSE A., GÉCZY J. Comparative study of pirlindole, a selective RIMA, and its two enantiomers using biochemical and behavioural techniques. Behav. Pharmacol. 1998;9:731–737. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

