De novo infection and serial transmission of Kaposi's sarcoma-associated herpesvirus in cultured endothelial cells
- PMID: 11836422
- PMCID: PMC153827
- DOI: 10.1128/jvi.76.5.2440-2448.2002
De novo infection and serial transmission of Kaposi's sarcoma-associated herpesvirus in cultured endothelial cells
Abstract
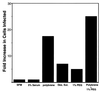
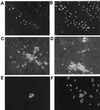
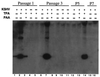
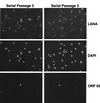
Infection by Kaposi's sarcoma-associated herpesvirus (KSHV) is central to the pathogenesis of the endothelial neoplasm Kaposi's sarcoma (KS) and is also linked to the rare B-cell tumor known as primary effusion lymphoma (PEL). Latently infected PEL cell lines can be induced to enter the lytic cycle and produce KSHV virions. However, such cells do not support de novo infection or serial propagation of KSHV. These limitations have prevented the development of systems for the genetic analysis of KSHV and have impeded a deeper understanding of KS pathogenesis. Here we show that human dermal microvascular endothelial cells immortalized by expression of telomerase can be readily infected by KSHV virions produced by PEL cells. Infection is predominantly latent, but a small subpopulation enters the lytic cycle spontaneously. Phorbol ester (tetradecanoyl phorbol acetate [TPA]) treatment of latently infected cells leads to enhanced induction of lytic KSHV replication, resulting in foci of cytopathic effect. There is no cytopathic effect or viral DNA expansion when infected TIME cells (telomerase-immortalized microvascular endothelial cells) are TPA induced in the presence of phosphonoacetic acid (PAA), an inhibitor of herpesvirus replication. Supernatants from phorbol-induced cultures transfer latent KSHV infection to uninfected cells, which can likewise be induced to undergo lytic replication by TPA treatment, and the virus can be further serially transmitted. Serial passage of the virus in TIME cells is completely inhibited when TPA treatment is done in the presence of PAA. Latently infected endothelial cells do not undergo major morphological changes or growth transformation, and infection is lost from the culture upon serial passage. This behavior faithfully recapitulates the behavior of spindle cells explanted from primary KS biopsies, strongly supporting the biological relevance of this culture system. These findings suggest that either the stability or the growth-deregulatory potential of the KSHV latency program in endothelial cells is more limited than might be predicted by analogy with other oncogenic viruses.
Figures


Similar articles
-
Spindle cell conversion by Kaposi's sarcoma-associated herpesvirus: formation of colonies and plaques with mixed lytic and latent gene expression in infected primary dermal microvascular endothelial cell cultures.J Virol. 2001 Jun;75(12):5614-26. doi: 10.1128/JVI.75.12.5614-5626.2001. J Virol. 2001. PMID: 11356969 Free PMC article.
-
Transcriptome analysis of Kaposi's sarcoma-associated herpesvirus during de novo primary infection of human B and endothelial cells.J Virol. 2015 Mar;89(6):3093-111. doi: 10.1128/JVI.02507-14. Epub 2014 Dec 31. J Virol. 2015. PMID: 25552714 Free PMC article.
-
Long-term-infected telomerase-immortalized endothelial cells: a model for Kaposi's sarcoma-associated herpesvirus latency in vitro and in vivo.J Virol. 2006 May;80(10):4833-46. doi: 10.1128/JVI.80.10.4833-4846.2006. J Virol. 2006. PMID: 16641275 Free PMC article.
-
[Replication Machinery of Kaposi's Sarcoma-associated Herpesvirus and Drug Discovery Research].Yakugaku Zasshi. 2019;139(1):69-73. doi: 10.1248/yakushi.18-00164-2. Yakugaku Zasshi. 2019. PMID: 30606932 Review. Japanese.
-
Viral latent proteins as targets for Kaposi's sarcoma and Kaposi's sarcoma-associated herpesvirus (KSHV/HHV-8) induced lymphoma.Curr Drug Targets Infect Disord. 2003 Jun;3(2):129-35. doi: 10.2174/1568005033481150. Curr Drug Targets Infect Disord. 2003. PMID: 12769790 Review.
Cited by
-
Latent KSHV Infected Endothelial Cells Are Glutamine Addicted and Require Glutaminolysis for Survival.PLoS Pathog. 2015 Jul 21;11(7):e1005052. doi: 10.1371/journal.ppat.1005052. eCollection 2015 Jul. PLoS Pathog. 2015. PMID: 26197457 Free PMC article.
-
Kaposi's sarcoma-associated herpesvirus virion-induced transcription activation of the ORF50 immediate-early promoter.J Virol. 2005 Oct;79(20):13180-5. doi: 10.1128/JVI.79.20.13180-13185.2005. J Virol. 2005. PMID: 16189019 Free PMC article.
-
Kaposi's Sarcoma-Associated Herpesvirus (KSHV)-Associated Disease in the AIDS Patient: An Update.Cancer Treat Res. 2019;177:63-80. doi: 10.1007/978-3-030-03502-0_3. Cancer Treat Res. 2019. PMID: 30523621 Free PMC article. Review.
-
Uncovering the complexities of Kaposi's sarcoma through genome-wide expression analysis.Genome Biol. 2004;5(11):247. doi: 10.1186/gb-2004-5-11-247. Epub 2004 Nov 1. Genome Biol. 2004. PMID: 15535873 Free PMC article. Review.
-
Surface downregulation of major histocompatibility complex class I, PE-CAM, and ICAM-1 following de novo infection of endothelial cells with Kaposi's sarcoma-associated herpesvirus.J Virol. 2003 Sep;77(17):9669-84. doi: 10.1128/jvi.77.17.9669-9684.2003. J Virol. 2003. PMID: 12915579 Free PMC article.
References
-
- Aluigi, M. G., A. Albini, S. Carlone, L. Repetto, R. De Marchi, A. Icardi, M. Moro, D. Noonan, and R. Benelli. 1996. KSHV sequences in biopsies and cultured spindle cells of epidemic, iatrogenic and Mediterranean forms of Kaposi's sarcoma. Res. Virol. 147:267-275. - PubMed
-
- Ballestas, M. E., P. A. Chatis, and K. M. Kaye. 1999. Efficient persistence of extrachromosomal KSHV DNA mediated by latency-associated nuclear antigen. Science 284:641-644. - PubMed
-
- Chang, Y., E. Cesarman, M. S. Pessin, F. Lee, J. Culpepper, D. M. Knowles, and P. S. Moore. 1994. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi's sarcoma. Science 266:1865-1869. - PubMed
-
- Ciufo, D. M., J. S. Cannon, L. J. Poole, F. Y. Wu, P. Murray, R. F. Ambinder, and G. S. Hayward. 2001. Spindle cell conversion by Kaposi's sarcoma-associated herpesvirus: formation of colonies and plaques with mixed lytic and latent gene expression in infected primary dermal microvascular endothelial cell cultures. J. Virol. 75:5614-5626. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

