Ca(2+) dynamics in the lumen of the endoplasmic reticulum in sensory neurons: direct visualization of Ca(2+)-induced Ca(2+) release triggered by physiological Ca(2+) entry
- PMID: 11847110
- PMCID: PMC125857
- DOI: 10.1093/emboj/21.4.622
Ca(2+) dynamics in the lumen of the endoplasmic reticulum in sensory neurons: direct visualization of Ca(2+)-induced Ca(2+) release triggered by physiological Ca(2+) entry
Abstract
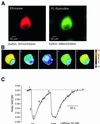
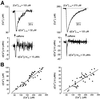
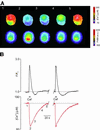
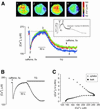
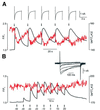
In cultured rat dorsal root ganglia neurons, we measured membrane currents, using the patch-clamp whole-cell technique, and the concentrations of free Ca(2+) in the cytosol ([Ca(2+)](i)) and in the lumen of the endoplasmic reticulum (ER) ([Ca(2+)](L)), using high- (Fluo-3) and low- (Mag-Fura-2) affinity Ca(2+)-sensitive fluorescent probes and video imaging. Resting [Ca(2+)](L) concentration varied between 60 and 270 microM. Activation of ryanodine receptors by caffeine triggered a rapid fall in [Ca(2+)](L) levels, which amounted to only 40--50% of the resting [Ca(2+)](L) value. Using electrophysiological depolarization, we directly demonstrate the process of Ca(2+)-induced Ca(2+) release triggered by Ca(2+) entry through voltage-gated Ca(2+) channels. The amplitude of Ca(2+) release from the ER lumen was linearly dependent on I(Ca).
Figures

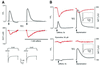

Similar articles
-
Neuronal endoplasmic reticulum acts as a single functional Ca2+ store shared by ryanodine and inositol-1,4,5-trisphosphate receptors as revealed by intra-ER [Ca2+] recordings in single rat sensory neurones.Pflugers Arch. 2003 Jul;446(4):447-54. doi: 10.1007/s00424-003-1094-z. Epub 2003 May 23. Pflugers Arch. 2003. PMID: 12764616
-
Fura-2 antagonises calcium-induced calcium release.Cell Calcium. 2003 Jan;33(1):27-35. doi: 10.1016/s0143-4160(02)00179-3. Cell Calcium. 2003. PMID: 12526885
-
Caffeine-induced calcium release from internal stores in cultured rat sensory neurons.Neuroscience. 1993 Dec;57(3):845-59. doi: 10.1016/0306-4522(93)90029-f. Neuroscience. 1993. PMID: 8309540
-
Interplay between ER Ca2+ uptake and release fluxes in neurons and its impact on [Ca2+] dynamics.Biol Res. 2004;37(4):665-74. doi: 10.4067/s0716-97602004000400024. Biol Res. 2004. PMID: 15709696 Review.
-
The endoplasmic reticulum as an integrating signalling organelle: from neuronal signalling to neuronal death.Eur J Pharmacol. 2002 Jul 5;447(2-3):141-54. doi: 10.1016/s0014-2999(02)01838-1. Eur J Pharmacol. 2002. PMID: 12151006 Review.
Cited by
-
Na+-dependent sources of intra-axonal Ca2+ release in rat optic nerve during in vitro chemical ischemia.J Neurosci. 2005 Oct 26;25(43):9960-7. doi: 10.1523/JNEUROSCI.2003-05.2005. J Neurosci. 2005. PMID: 16251444 Free PMC article.
-
TALK-1 channels control β cell endoplasmic reticulum Ca2+ homeostasis.Sci Signal. 2017 Sep 19;10(497):eaan2883. doi: 10.1126/scisignal.aan2883. Sci Signal. 2017. PMID: 28928238 Free PMC article.
-
Ca2+ and mitochondria as substrates for deficits in synaptic plasticity in normal brain ageing.J Cell Mol Med. 2004 Apr-Jun;8(2):181-90. doi: 10.1111/j.1582-4934.2004.tb00273.x. J Cell Mol Med. 2004. PMID: 15256066 Free PMC article. Review.
-
From Galvani to patch clamp: the development of electrophysiology.Pflugers Arch. 2006 Dec;453(3):233-47. doi: 10.1007/s00424-006-0169-z. Epub 2006 Oct 28. Pflugers Arch. 2006. PMID: 17072639
-
Endoplasmic reticulum Ca(2+) handling in excitable cells in health and disease.Pharmacol Rev. 2011 Sep;63(3):700-27. doi: 10.1124/pr.110.003814. Epub 2011 Jul 7. Pharmacol Rev. 2011. PMID: 21737534 Free PMC article. Review.
References
-
- Berridge M.J. (1993) Inositoltrisphosphate and calcium signalling. Nature, 361, 315–325. - PubMed
-
- Berridge M.J. (1998) Neuronal calcium signaling. Neuron, 21, 13–26. - PubMed
-
- Bezprozvanny I., Watras,J. and Ehrlich,B.E. (1991) Bell-shaped calcium-response curves of Ins(1,4,5)P3- and calcium-gated channels from endoplasmic reticulum of cerebellum. Nature, 351, 751–754. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

