Plasma protein binding of amphotericin B and pharmacokinetics of bound versus unbound amphotericin B after administration of intravenous liposomal amphotericin B (AmBisome) and amphotericin B deoxycholate
- PMID: 11850269
- PMCID: PMC127463
- DOI: 10.1128/AAC.46.3.834-840.2002
Plasma protein binding of amphotericin B and pharmacokinetics of bound versus unbound amphotericin B after administration of intravenous liposomal amphotericin B (AmBisome) and amphotericin B deoxycholate
Abstract
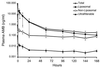
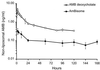
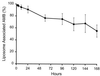
Unilamellar liposomal amphotericin B (AmBisome) (liposomal AMB) reduces the toxicity of this antifungal drug. The unique composition of liposomal AMB stabilizes the liposomes, producing higher sustained drug levels in plasma and reducing renal and hepatic excretion. When liposomes release their drug payload, unbound, protein-bound, and liposomal drug pools may exist simultaneously in the body. To determine the amounts of drug in these pools, we developed a procedure to measure unbound AMB in human plasma by ultrafiltration and then used it to characterize AMB binding in vitro and to assess the pharmacokinetics of nonliposomal pools of AMB in a phase IV study of liposomal AMB and AMB deoxycholate in healthy subjects. We confirmed that AMB is highly bound (>95%) in human plasma and showed that both human serum albumin and alpha(1)-acid glycoprotein contribute to this binding. AMB binding exhibited an unusual concentration dependence in plasma: the percentage of bound drug increased as the AMB concentration increased. This was attributed to the low solubility of AMB in plasma, which limits the unbound drug concentration to <1 microg/ml. Subjects given 2 mg of liposomal AMB/kg of body weight had lower exposures (as measured by the maximum concentration of drug in serum and the area under the concentration-time curve) to both unbound and nonliposomal drug than those receiving 0.6 mg of AMB deoxycholate/kg. Most of the AMB in plasma remained liposome associated (97% at 4 h, 55% at 168 h) after liposomal AMB administration, so that unbound drug concentrations remained at <25 ng/ml in all liposomal AMB-treated subjects. Although liposomal AMB markedly reduces the total urinary and fecal recoveries of AMB, urinary and fecal clearances based on unbound AMB were similar (94 to 121 ml h(-1) kg(-1)) for both formulations. Unbound drug urinary clearances were equal to the glomerular filtration rate, and tubular transit rates were <16% of the urinary excretion rate, suggesting that net filtration of unbound drug, with little secretion or reabsorption, is the mechanism of renal clearance for both conventional and liposomal AMB in humans. Unbound drug fecal clearances were also similar for the two formulations. Thus, liposomal AMB increases total AMB concentrations while decreasing unbound AMB concentrations in plasma as a result of sequestration of the drug in long-circulating liposomes.
Figures

References
-
- Adler-Moore, J., and R. T. Proffitt. 1998. AmBisome: long circulating liposomal formulation of amphotericin B, p. 185-206. In M. C. Woodle and G. Storm (ed.), Long-circulating liposomes: old drugs, new therapeutics. Landes Bioscience, Georgetown, Tex.
-
- Alak, A., S. Moys, and I. Bekersky. 1996. A high-performance liquid chromatographic assay for the determination of amphotericin B serum concentrations after the administration of AmBisome, a liposomal amphotericin B formulation. Ther. Drug Monit. 18:604-609. - PubMed
-
- Bekersky, I., and W. A. Colburn. 1981. Renal clearance of carprofen in the isolated perfused rat kidney. Drug Metab. Dispos. 9:25-29. - PubMed
-
- Bekersky, I., R. M. Fielding, D. Buell, and I. Lawrence. 1999. Lipid-based amphotericin B formulations: from animals to man. Pharm. Sci. Technol. Today 2:230-236. - PubMed
-
- Bekersky, I., D. Buell, M. Tomishima, K. Maki, I. Lawrence, and R. M. Fielding. 1999. New approaches to systemic antifungal therapy: case studies of AmBisome and FK463. Recent Res. Dev. Antimicrob. Agents Chemother. 3:407-413.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

