Prevention of a hypoxic Ca(2+)(i) response by SERCA inhibitors in cerebral arterioles
- PMID: 11861320
- PMCID: PMC1573211
- DOI: 10.1038/sj.bjp.0704547
Prevention of a hypoxic Ca(2+)(i) response by SERCA inhibitors in cerebral arterioles
Abstract
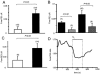
1. The aim of the study was to investigate the mechanism of a novel effect of hypoxia on intracellular Ca(2+) signalling in rabbit cerebral arteriolar smooth muscle cells, an effect that was resistant to the L-type Ca(2+) channel antagonist methoxyverapamil (D600). 2.[Ca(2+)](i) of smooth muscle cells in intact arteriolar fragments was measured using the Ca(2+)-indicator dye fura-PE3. Hypoxia (PO(2) 10 - 20 mmHg) lowered basal [Ca(2+)](i) but did not inhibit Ca(2+) entry pathways measured by Mn(2+)-quenching of fura-PE3. 3. The effect of hypoxia was completely prevented by thapsigargin or cyclopiazonic acid, selective inhibitors of sarcoplasmic reticulum Ca(2+) ATPase (SERCA). Since these inhibitors do not block Ca(2+) extrusion or uptake via the plasma membrane, the data indicate that the effect of hypoxia depends on a functional sarcoplasmic reticulum. 4. Because actions of nitric oxide (NO) on vascular smooth muscle are also prevented by SERCA inhibitors it was explored whether the effect of hypoxia occurred via modulation of endogenous NO release. Residual NOS-I and NOS-III were detected by immunostaining, and there were NO-dependent effects of NOS inhibitors on Ca(2+)(i)-signalling. Nevertheless, inhibition of endogenous NO production did not prevent the effect of hypoxia on [Ca(2+)](i). 5. The experiments reveal a novel nitric oxide-independent effect of hypoxia that is prevented by SERCA inhibitors.
Figures




References
-
- ASHWAL S., PEARCE W.J., IGNARRO L.J. Hypoxia increases cGMP and decreases calcium uptake in rabbit cranial arteries. Proc. West Pharmacol. Soc. 1988;31:125–128. - PubMed
-
- BEECH D.J. Actions of neurotransmitters and other messengers on Ca2+ channels and K+ channels in smooth muscle cells. Pharmacology and Therapeutics. 1997;73:91–119. - PubMed
-
- BEECH D.J., CHEONG A., FLEMMING R., GUIBERT C., XU S.Z.Modulation of vascular K+ channels by extracellular messengers Potassium Channels in Cardiovascular Biology>” 2001New York: Kluwer Academic/Plenum Publishers; 457–476.eds. Archer, S.L. & Rusch, N.J. Chapter 23. pp
-
- BERNE R.M., RUBIO R., CURNISH R.R. Release of adenosine from ischemic brain: Effect on cerebral vascular resistance and incorporation into cerebral adenine nucleotides. Circ. Res. 1974;35:262–271.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

