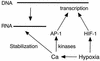
The regulation of hypoxic genes by calcium involves c-Jun/AP-1, which cooperates with hypoxia-inducible factor 1 in response to hypoxia
- PMID: 11865053
- PMCID: PMC135615
- DOI: 10.1128/MCB.22.6.1734-1741.2002
The regulation of hypoxic genes by calcium involves c-Jun/AP-1, which cooperates with hypoxia-inducible factor 1 in response to hypoxia
Abstract
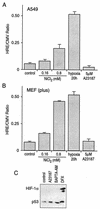
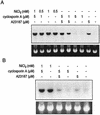
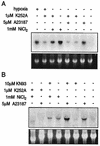
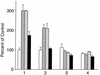
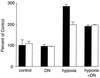
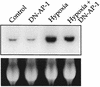
Hypoxia causes the accumulation of the transcription factor hypoxia-inducible factor 1 (HIF-1), culminating in the expression of hypoxia-inducible genes such as those for vascular endothelial growth factor (VEGF) and NDRG-1/Cap43. Previously, we have demonstrated that intracellular calcium (Ca(2+)) is required for the expression of hypoxia-inducible genes. Here we found that, unlike with hypoxia or hypoxia-mimicking conditions, the elevation of intracellular Ca(2+) neither induced the HIF-1alpha protein nor stimulated HIF-1-dependent transcription. Furthermore, the elevation of intracellular Ca(2+) induced NDRG-1/Cap43 mRNA in HIF-1alpha-deficient cells. It also increased levels of c-Jun protein, causing its phosphorylation. The protein kinase inhibitor K252a abolished c-Jun induction and activator protein 1 (AP-1)-dependent reporter expression caused by Ca(2+) ionophore or hypoxia. K252a also significantly decreased hypoxia-induced VEGF and NDRG-1/Cap43 gene expression in both human and mouse cells. Using a set of deletion VEGF-Luc promoter constructs, we found that both HIF-1 and two AP-1 sites contribute to hypoxia-mediated induction of transcription. In contrast, only AP-1 sites contributed to Ca(2+)-mediated VEGF-Luc induction. A dominant-negative AP-1 prevented Ca(2+)-dependent transcription and partially impaired hypoxia-mediated transcription. In addition, dominant-negative AP-1 diminished the expression of the NDRG-1/Cap43 gene following hypoxia. We conclude that during hypoxia, an increase in intracellular Ca(2+) activates a HIF-1-independent signaling pathway that involves AP-1-dependent transcription. Cooperation between the HIF-1 and AP-1 pathways allows fine regulation of gene expression during hypoxia.
Figures



References
-
- An, W. G., M. Kanekal, M. C. Simon, E. Maltepe, M. V. Blagosklonny, and L. M. Neckers. 1998. Stabilization of wild-type p53 by hypoxia-inducible factor 1α. Nature 392:405-408. - PubMed
-
- Arnould, T., C. Michiels, I. Alexandre, and J. Remacle. 1992. Effect of hypoxia upon intracellular calcium concentration of human endothelial cells. J. Cell. Physiol. 152:215-221. - PubMed
-
- Berra, E., J. Milanini, D. E. Richard, M. Le Gall, F. Vinals, E. Gothie, D. Roux, G. Pages, and J. Pouyssegur. 2000. Signaling angiogenesis via p42/p44 MAP kinase and hypoxia. Biochem. Pharmacol. 60:1171-1178. - PubMed
-
- Chabannes, E., S. Fauconnet, S. Bernardini, H. Wallerand, G. Adessi, and H. Bittard. 2001. Protein kinase C signalling pathway is involved in the regulation of vascular endothelial growth factor expression in human bladder transitional carcinoma cells. Cell. Signal. 13:585-591. - PubMed
-
- Chen, C. Y., F. Del Gatto-Konczak, Z. Wu, and M. Karin. 1998. Stabilization of interleukin-2 mRNA by the c-Jun NH2-terminal kinase pathway. Science 280:1945-1949. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
