Characterization of an apamin-sensitive small-conductance Ca(2+)-activated K(+) channel in porcine coronary artery endothelium: relevance to EDHF
- PMID: 11877319
- PMCID: PMC1573217
- DOI: 10.1038/sj.bjp.0704551
Characterization of an apamin-sensitive small-conductance Ca(2+)-activated K(+) channel in porcine coronary artery endothelium: relevance to EDHF
Abstract
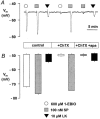
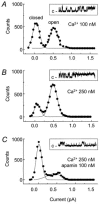
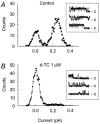
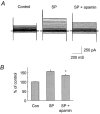
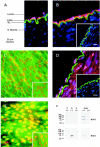
1. The apamin-sensitive small-conductance Ca(2+)-activated K(+) channel (SK(Ca)) was characterized in porcine coronary arteries. 2. In intact arteries, 100 nM substance P and 600 microM 1-ethyl-2-benzimidazolinone (1-EBIO) produced endothelial cell hyperpolarizations (27.8 +/- 0.8 mV and 24.1 +/- 1.0 mV, respectively). Charybdotoxin (100 nM) abolished the 1-EBIO response but substance P continued to induce a hyperpolarization (25.8 +/- 0.3 mV). 3. In freshly-isolated endothelial cells, outside-out patch recordings revealed a unitary K(+) conductance of 6.8 +/- 0.04 pS. The open-probability was increased by Ca(2+) and reduced by apamin (100 nM). Substance P activated an outward current under whole-cell perforated-patch conditions and a component of this current (38%) was inhibited by apamin. A second conductance of 2.7 +/- 0.03 pS inhibited by d-tubocurarine was observed infrequently. 4. Messenger RNA encoding the SK2 and SK3, but not the SK1, subunits of SK(Ca) was detected by RT - PCR in samples of endothelium. Western blotting indicated that SK3 protein was abundant in samples of endothelium compared to whole arteries. SK2 protein was present in whole artery nuclear fractions. 5. Immunofluorescent labelling confirmed that SK3 was highly expressed at the plasmalemma of endothelial cells and was not expressed in smooth muscle. SK2 was restricted to the peri-nuclear regions of both endothelial and smooth muscle cells. 6. In conclusion, the porcine coronary artery endothelium expresses an apamin-sensitive SK(Ca) containing the SK3 subunit. These channels are likely to confer all or part of the apamin-sensitive component of the endothelium-derived hyperpolarizing factor (EDHF) response.
Figures


References
-
- BARFOD E.T., MOORE A.L., LIDOFSKY S.D. Cloning and functional expression of a liver isoform of the small conductance Ca2+-activated K+ channel SK3. Am. J. Physiol. 2001;280:C836–C842. - PubMed
-
- BRADFORD M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilising the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. - PubMed
-
- BURNHAM M.P., RICHARDS G.R., EDWARDS G., WESTON A.H.SK3 Ca2+-activated K+ channels are located on endothelia that generate apamin-sensitive EDHF responses Proc. Western Pharmacol. Soc. 2001(In press)
-
- BYCHKOV R., GOLLASCH M., RIED C., LUFT F.C., HALLER H. Effects of pinacidil on K+ channels in human coronary artery vascular smooth muscle cells. Am. J. Physiol. 1997;273:C161–C171. - PubMed
-
- CAO Y.-J., DREIXLER J.C., ROIZEN J.D., ROBERTS M.T., HOUAMED K.M. Modulation of recombinant small-conductance Ca2+-activated K+ channels by the muscle relaxant chlorzoxazone and structurally related compounds. J. Pharmacol. Exp. Ther. 2001;296:683–689. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

