Essential immunoregulatory role for BCAP in B cell development and function
- PMID: 11877477
- PMCID: PMC2193770
- DOI: 10.1084/jem.20011751
Essential immunoregulatory role for BCAP in B cell development and function
Abstract
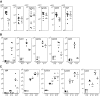
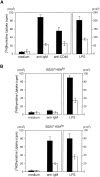
BCAP was recently cloned as a binding molecule to phosphoinositide 3-kinase (PI3K). To investigate the role of BCAP, mutant mice deficient in BCAP were generated. While BCAP-deficient mice are viable, they have decreased numbers of mature B cells and B1 B cell deficiency. The mice produce lower titers of serum immunoglobulin (Ig)M and IgG3, and mount attenuated responses to T cell--independent type II antigen. Upon B cell receptor cross-linking, BCAP-deficient B cells exhibit reduced Ca(2+) mobilization and poor proliferative responses. These findings demonstrate that BCAP plays a pivotal immunoregulatory role in B cell development and humoral immune responses.
Figures
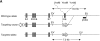
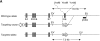


References
-
- Meffre, E., R. Casellas, and M.C. Nussenzweig. 2000. Antibody regulation of B cell development. Nat. Immunol. 1:379–385. - PubMed
-
- Hardy, R.R., and K. Hayakawa. 2001. B cell development pathways. Annu. Rev. Immunol. 19:595–621. - PubMed
-
- Benschop, R.J., and J.C. Cambier. 1999. B cell development: signal transduction by antigen receptors and their surrogates. Curr. Opin. Immunol. 11:143–151. - PubMed
-
- Rajewsky, K. 1996. Clonal selection and learning in the antibody system. Nature. 381:751–758. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

