The effects of temperature on vesicular supply and release in autaptic cultures of rat and mouse hippocampal neurons
- PMID: 11882684
- PMCID: PMC2290147
- DOI: 10.1113/jphysiol.2001.013277
The effects of temperature on vesicular supply and release in autaptic cultures of rat and mouse hippocampal neurons
Abstract
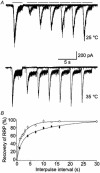
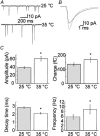
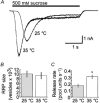
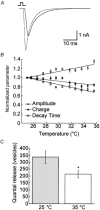
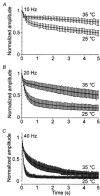
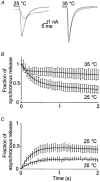
Membrane fusion plays a central role in the synaptic vesicle cycle. While many of the pre- and postfusion events have been investigated at room temperature, few researchers have investigated these processes at more physiologically relevant temperatures. We have used autaptic cultures of hippocampal neurons to investigate changes in the size and refilling rate of the readily releasable pool (RRP) of synaptic vesicles brought about by an increase in temperature from 25 to 35 degrees C. We have also examined temperature-dependent changes in spontaneous and action potential (AP)-evoked release as well as the fraction of the RRP that is released during an AP. Although we found a threefold increase in the refilling rate of the RRP at the higher temperature, there was no apparent change in the size of the RRP with increased temperature. Moreover, we observed a slight but significant decrease in the quanta released during an AP. This increased refilling rate and decreased release probability resulted in a reduction of both the degree and time course of synaptic depression during high frequency stimulation at the higher temperature. This reduction in synaptic depression was accompanied by an increased maintenance of the synchronous component of release during high frequency stimulation. These findings indicate that the dynamics of vesicular supply and release in hippocampal neurons at room temperature are significantly different at near physiological temperatures and could affect our present understanding of the way in which individual neurons and networks of neurons process information.
Figures
Similar articles
-
Autapses and networks of hippocampal neurons exhibit distinct synaptic transmission phenotypes in the absence of synaptotagmin I.J Neurosci. 2009 Jun 10;29(23):7395-403. doi: 10.1523/JNEUROSCI.1341-09.2009. J Neurosci. 2009. PMID: 19515907 Free PMC article.
-
Reluctant vesicles contribute to the total readily releasable pool in glutamatergic hippocampal neurons.J Neurosci. 2005 Apr 13;25(15):3842-50. doi: 10.1523/JNEUROSCI.5231-04.2005. J Neurosci. 2005. PMID: 15829636 Free PMC article.
-
BDNF increases release probability and the size of a rapidly recycling vesicle pool within rat hippocampal excitatory synapses.J Physiol. 2006 Aug 1;574(Pt 3):787-803. doi: 10.1113/jphysiol.2006.111310. Epub 2006 May 18. J Physiol. 2006. PMID: 16709633 Free PMC article.
-
Competition between phasic and asynchronous release for recovered synaptic vesicles at developing hippocampal autaptic synapses.J Neurosci. 2004 Jan 14;24(2):420-33. doi: 10.1523/JNEUROSCI.4452-03.2004. J Neurosci. 2004. PMID: 14724240 Free PMC article.
-
Excitatory synaptic transmission is depressed in cultured hippocampal neurons of APP/PS1 mice.Neurobiol Aging. 2009 Aug;30(8):1227-37. doi: 10.1016/j.neurobiolaging.2007.10.016. Epub 2008 Feb 20. Neurobiol Aging. 2009. PMID: 18077058
Cited by
-
Dynamics of the readily releasable pool during post-tetanic potentiation in the rat calyx of Held synapse.J Physiol. 2007 Jun 1;581(Pt 2):467-78. doi: 10.1113/jphysiol.2006.127365. Epub 2007 Mar 15. J Physiol. 2007. PMID: 17363387 Free PMC article.
-
Dynamic Recovery from Depression Enables Rate Encoding in Inhibitory Synapses.iScience. 2020 Mar 27;23(3):100940. doi: 10.1016/j.isci.2020.100940. Epub 2020 Feb 27. iScience. 2020. PMID: 32163896 Free PMC article.
-
How to Build a Fast and Highly Sensitive Sound Detector That Remains Robust to Temperature Shifts.J Neurosci. 2019 Sep 11;39(37):7260-7276. doi: 10.1523/JNEUROSCI.2510-18.2019. Epub 2019 Jul 17. J Neurosci. 2019. PMID: 31315946 Free PMC article.
-
Additive effects on the energy barrier for synaptic vesicle fusion cause supralinear effects on the vesicle fusion rate.Elife. 2015 Apr 14;4:e05531. doi: 10.7554/eLife.05531. Elife. 2015. PMID: 25871846 Free PMC article.
-
Hippocampal neurons in direct contact with astrocytes exposed to amyloid β25-35 exhibit reduced excitatory synaptic transmission.IBRO Rep. 2019 Jul 23;7:34-41. doi: 10.1016/j.ibror.2019.07.1719. eCollection 2019 Dec. IBRO Rep. 2019. PMID: 31388597 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

