Redirecting retroviral tropism by insertion of short, nondisruptive peptide ligands into envelope
- PMID: 11884580
- PMCID: PMC136016
- DOI: 10.1128/jvi.76.7.3558-3563.2002
Redirecting retroviral tropism by insertion of short, nondisruptive peptide ligands into envelope
Abstract
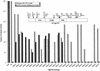
A potentially powerful approach for in vivo gene delivery is to target retrovirus to specific cells through interactions between cell surface receptors and appropriately modified viral envelope proteins. Previously, relatively large (>100 residues) protein ligands to cell surface receptors have been inserted at or near the N terminus of retroviral envelope proteins. Although viral tropism could be altered, the chimeric envelope proteins lacked full activity, and coexpression of wild-type envelope was required for production of transducing virus. Here we analyze more than 40 derivatives of ecotropic Moloney murine leukemia virus (MLV) envelope, containing insertions of short RGD-containing peptides, which are ligands for integrin receptors. In many cases pseudotyped viruses containing only the chimeric envelope protein could transduce human cells. The precise location, size, and flanking sequences of the ligand affected transduction specificity and efficiency. We conclude that retroviral tropism can be rationally reengineered by insertion of short peptide ligands and without the need to coexpress wild-type envelope.
Figures

Similar articles
-
Redirecting retroviral tropism by insertion of hepatitis B virus PreS1 core peptide into the envelope.Arch Virol. 2007;152(9):1721-30. doi: 10.1007/s00705-007-0987-0. Epub 2007 May 24. Arch Virol. 2007. PMID: 17520321
-
Selective targeting and inducible destruction of human cancer cells by retroviruses with envelope proteins bearing short peptide ligands.J Virol. 2002 Apr;76(7):3564-9. doi: 10.1128/jvi.76.7.3564-3569.2002. J Virol. 2002. PMID: 11884581 Free PMC article.
-
Identification of regions in the Moloney murine leukemia virus SU protein that tolerate the insertion of an integrin-binding peptide.Virology. 2000 Mar 30;269(1):7-17. doi: 10.1006/viro.2000.0201. Virology. 2000. PMID: 10725193
-
Cell targeting by murine recombinant retroviruses.Bone Marrow Transplant. 1992;9 Suppl 1:139-42. Bone Marrow Transplant. 1992. PMID: 1504656 Review.
-
Herpesvirus/retrovirus chimeric vectors.Curr Gene Ther. 2004 Dec;4(4):409-16. doi: 10.2174/1566523043345922. Curr Gene Ther. 2004. PMID: 15578990 Review.
Cited by
-
Polysaccharide-modified scaffolds for controlled lentivirus delivery in vitro and after spinal cord injury.J Control Release. 2013 Sep 28;170(3):421-9. doi: 10.1016/j.jconrel.2013.06.013. Epub 2013 Jun 18. J Control Release. 2013. PMID: 23791981 Free PMC article.
-
Molecular Engineering of Virus Tropism.Int J Mol Sci. 2024 Oct 15;25(20):11094. doi: 10.3390/ijms252011094. Int J Mol Sci. 2024. PMID: 39456875 Free PMC article. Review.
-
Change of tropism of SL3-2 murine leukemia virus, using random mutational libraries.J Virol. 2004 Sep;78(17):9343-51. doi: 10.1128/JVI.78.17.9343-9351.2004. J Virol. 2004. PMID: 15308729 Free PMC article.
-
Molecular engineering of viral gene delivery vehicles.Annu Rev Biomed Eng. 2008;10:169-94. doi: 10.1146/annurev.bioeng.10.061807.160514. Annu Rev Biomed Eng. 2008. PMID: 18647114 Free PMC article. Review.
-
Empowering adult stem cells for myocardial regeneration.Circ Res. 2011 Dec 9;109(12):1415-28. doi: 10.1161/CIRCRESAHA.111.243071. Circ Res. 2011. PMID: 22158649 Free PMC article. Review.
References
-
- Albritton, L. M., L. Tseng, D. Scadden, and J. M. Cunningham. 1989. A putative murine ecotropic retrovirus receptor gene encodes a multiple membrane-spanning protein and confers susceptibility to virus infection. Cell 57:659-666. - PubMed
-
- Allman, R., P. Cowburn, and M. Mason. 2000. In vitro and in vivo effects of a cyclic peptide with affinity for the ανβ3 integrin in human melanoma cells. Eur. J. Cancer 36:410-422. - PubMed
-
- Aumailley, M., M. Gerl, A. Sonnenberg, R. Deutzmann, and R. Timpl. 1990. Identification of the Arg-Gly-Asp sequence in laminin A chain as a latent cell-binding site being exposed in fragment P1. FEBS Lett. 262:82-86. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

