Promoter-specific functions of CIITA and the MHC class II enhanceosome in transcriptional activation
- PMID: 11889043
- PMCID: PMC125922
- DOI: 10.1093/emboj/21.6.1379
Promoter-specific functions of CIITA and the MHC class II enhanceosome in transcriptional activation
Abstract
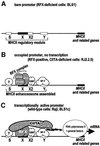
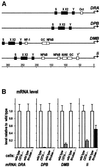
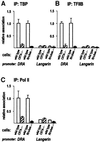
Transcription of the major histocompatibility complex class II family of genes is regulated by conserved promoter elements and two gene-specific trans-activators, RFX and CIITA. RFX binds DNA and nucleates the assembly of an enhanceosome, which recruits CIITA through protein--protein interactions. Transcriptional activation is a complex, multi-step process involving chromatin modification and recruitment of the transcription apparatus. To examine the roles of the enhanceosome and CIITA in these processes, we analysed the level of promoter-associated hyperacetylated histones H3 and H4, TBP, TFIIB and RNA poly merase II in cells lacking RFX or CIITA. We compared four genes co-regulated by RFX and CIITA (HLA-DRA, HLA-DPB, HLA-DMB and Ii) and found that the enhanceosome and CIITA make variable, promoter-dependent contributions to histone acetylation and transcription apparatus recruitment. CIITA is generally implicated at multiple levels of the activation process, while the enhanceosome contributes in a CIITA-independent manner only at certain promoters. Our results support the general notion that the impact of a particular activator on transcription in vivo may vary depending on the promoter and the chromatin context.
Figures

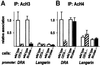

Similar articles
-
Kaposi's Sarcoma-Associated Herpesvirus Latency-Associated Nuclear Antigen Inhibits Major Histocompatibility Complex Class II Expression by Disrupting Enhanceosome Assembly through Binding with the Regulatory Factor X Complex.J Virol. 2015 May;89(10):5536-56. doi: 10.1128/JVI.03713-14. Epub 2015 Mar 4. J Virol. 2015. PMID: 25740990 Free PMC article.
-
The transcription factor RFX protects MHC class II genes against epigenetic silencing by DNA methylation.J Immunol. 2009 Aug 15;183(4):2545-53. doi: 10.4049/jimmunol.0900376. Epub 2009 Jul 20. J Immunol. 2009. PMID: 19620312
-
Long distance control of MHC class II expression by multiple distal enhancers regulated by regulatory factor X complex and CIITA.J Immunol. 2004 Nov 15;173(10):6200-10. doi: 10.4049/jimmunol.173.10.6200. J Immunol. 2004. PMID: 15528357
-
Lessons from the bare lymphocyte syndrome: molecular mechanisms regulating MHC class II expression.Immunol Rev. 2000 Dec;178:148-65. doi: 10.1034/j.1600-065x.2000.17813.x. Immunol Rev. 2000. PMID: 11213800 Review.
-
A common set of factors control the expression of the MHC class II, invariant chain, and HLA-DM genes.Microbes Infect. 1999 Sep;1(11):847-53. doi: 10.1016/s1286-4579(99)00234-8. Microbes Infect. 1999. PMID: 10614001 Review. No abstract available.
Cited by
-
c-Rel-dependent monocytes are potent immune suppressor cells in cancer.J Leukoc Biol. 2022 Oct;112(4):845-859. doi: 10.1002/JLB.1MA0422-518RR. Epub 2022 Jun 13. J Leukoc Biol. 2022. PMID: 35694784 Free PMC article.
-
Identification of CIITA regulated genetic module dedicated for antigen presentation.PLoS Genet. 2008 Apr 25;4(4):e1000058. doi: 10.1371/journal.pgen.1000058. PLoS Genet. 2008. PMID: 18437201 Free PMC article.
-
Blocking MHC class II on human endothelium mitigates acute rejection.JCI Insight. 2016;1(1):e85293. doi: 10.1172/jci.insight.85293. Epub 2016 Jan 21. JCI Insight. 2016. PMID: 26900601 Free PMC article.
-
Combination of the histone deacetylase inhibitor depsipeptide and 5-fluorouracil upregulates major histocompatibility complex class II and p21 genes and activates caspase-3/7 in human colon cancer HCT-116 cells.Oncol Rep. 2016 Oct;36(4):1875-85. doi: 10.3892/or.2016.5008. Epub 2016 Aug 8. Oncol Rep. 2016. PMID: 27509880 Free PMC article.
-
New functions of the major histocompatibility complex class II-specific transcription factor RFXANK revealed by a high-resolution mutagenesis study.Mol Cell Biol. 2005 Oct;25(19):8607-18. doi: 10.1128/MCB.25.19.8607-8618.2005. Mol Cell Biol. 2005. PMID: 16166641 Free PMC article.
References
-
- Alfonso C. and Karlsson,L. (2000) Nonclassical MHC class II molecules. Annu. Rev. Immunol., 18, 113–142. - PubMed
-
- Beresford G.W. and Boss,J.M. (2001) CIITA coordinates multiple histone acetylation modifications at the HLA-DRA promoter. Nature Immunol., 2, 652–657. - PubMed
-
- Brown A.M., Barr,C.L. and Ting,J.P. (1991) Sequences homologous to class II MHC W, X and Y elements mediate constitutive and IFN-γ-induced expression of human class II-associated invariant chain gene. J. Immunol., 146, 3183–3189. - PubMed
-
- Brown A.M., Linhoff,M.W., Stein,B., Wright,K.L., Baldwin,A.S.,Jr, Basta,P.V. and Ting,J.P. (1994) Function of NF-κB/Rel binding sites in the major histocompatibility complex class II invariant chain promoter is dependent on cell-specific binding of different NF-κB/Rel subunits. Mol. Cell. Biol., 14, 2926–2935. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

