Chemiosmotic energy conservation with Na(+) as the coupling ion during hydrogen-dependent caffeate reduction by Acetobacterium woodii
- PMID: 11889102
- PMCID: PMC134933
- DOI: 10.1128/JB.184.7.1947-1951.2002
Chemiosmotic energy conservation with Na(+) as the coupling ion during hydrogen-dependent caffeate reduction by Acetobacterium woodii
Abstract
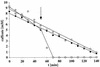
Cell suspensions of Acetobacterium woodii prepared from cultures grown on fructose plus caffeate catalyzed caffeate reduction with electrons derived from molecular hydrogen. Hydrogen-dependent caffeate reduction was strictly Na(+) dependent with a K(m) for Na(+) of 0.38 mM; Li(+) could substitute for Na(+). The sodium ionophore ETH2120, but not protonophores, stimulated hydrogen-dependent caffeate reduction by 280%, indicating that caffeate reduction is coupled to the buildup of a membrane potential generated by primary Na(+) extrusion. Caffeate reduction was coupled to the synthesis of ATP, and again, ATP synthesis coupled to hydrogen-dependent caffeate reduction was strictly Na(+) dependent and abolished by ETH2120, but not by protonophores, indicating the involvement of a transmembrane Na(+) gradient in ATP synthesis. The ATPase inhibitor N,N'-dicyclohexylcarbodiimide (DCCD) abolished ATP synthesis, and at the same time, hydrogen-dependent caffeate reduction was inhibited. This inhibition could be relieved by ETH2120. These experiments are fully compatible with a chemiosmotic mechanism of ATP synthesis with Na(+) as the coupling ion during hydrogen-dependent caffeate reduction by A. woodii.
Figures
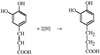



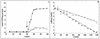

Similar articles
-
Caffeate respiration in the acetogenic bacterium Acetobacterium woodii: a coenzyme A loop saves energy for caffeate activation.Appl Environ Microbiol. 2013 Mar;79(6):1942-7. doi: 10.1128/AEM.03604-12. Epub 2013 Jan 11. Appl Environ Microbiol. 2013. PMID: 23315745 Free PMC article.
-
Dissection of the caffeate respiratory chain in the acetogen Acetobacterium woodii: identification of an Rnf-type NADH dehydrogenase as a potential coupling site.J Bacteriol. 2007 Nov;189(22):8145-53. doi: 10.1128/JB.01017-07. Epub 2007 Sep 14. J Bacteriol. 2007. PMID: 17873051 Free PMC article.
-
Regulation of caffeate respiration in the acetogenic bacterium Acetobacterium woodii.Appl Environ Microbiol. 2007 Jun;73(11):3630-6. doi: 10.1128/AEM.02060-06. Epub 2007 Apr 6. Appl Environ Microbiol. 2007. PMID: 17416687 Free PMC article.
-
The ins and outs of Na(+) bioenergetics in Acetobacterium woodii.Biochim Biophys Acta. 2009 Jun;1787(6):691-6. doi: 10.1016/j.bbabio.2008.12.015. Epub 2009 Jan 8. Biochim Biophys Acta. 2009. PMID: 19167341 Review.
-
The Na(+) cycle in Acetobacterium woodii: identification and characterization of a Na(+) translocating F(1)F(0)-ATPase with a mixed oligomer of 8 and 16 kDa proteolipids.Biochim Biophys Acta. 2001 May 1;1505(1):108-20. doi: 10.1016/s0005-2728(00)00281-4. Biochim Biophys Acta. 2001. PMID: 11248193 Review.
Cited by
-
Caffeate respiration in the acetogenic bacterium Acetobacterium woodii: a coenzyme A loop saves energy for caffeate activation.Appl Environ Microbiol. 2013 Mar;79(6):1942-7. doi: 10.1128/AEM.03604-12. Epub 2013 Jan 11. Appl Environ Microbiol. 2013. PMID: 23315745 Free PMC article.
-
The Rnf complex of Clostridium ljungdahlii is a proton-translocating ferredoxin:NAD+ oxidoreductase essential for autotrophic growth.mBio. 2012 Dec 26;4(1):e00406-12. doi: 10.1128/mBio.00406-12. mBio. 2012. PMID: 23269825 Free PMC article.
-
Overcoming Energetic Barriers in Acetogenic C1 Conversion.Front Bioeng Biotechnol. 2020 Dec 23;8:621166. doi: 10.3389/fbioe.2020.621166. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 33425882 Free PMC article. Review.
-
Biochemistry, evolution and physiological function of the Rnf complex, a novel ion-motive electron transport complex in prokaryotes.Cell Mol Life Sci. 2011 Feb;68(4):613-34. doi: 10.1007/s00018-010-0555-8. Epub 2010 Nov 12. Cell Mol Life Sci. 2011. PMID: 21072677 Free PMC article. Review.
-
Functional production of the Na+ F1F(O) ATP synthase from Acetobacterium woodii in Escherichia coli requires the native AtpI.J Bioenerg Biomembr. 2013 Feb;45(1-2):15-23. doi: 10.1007/s10863-012-9474-8. Epub 2012 Oct 3. J Bioenerg Biomembr. 2013. PMID: 23054076
References
-
- Aufurth, S., H. Schägger, and V. Müller. 2000. Identification of subunits a, b, and c1 from Acetobacterium woodii Na+-F1F0-ATPase. Subunits c1, c2, and c3 constitute a mixed c-oligomer. J. Biol. Chem. 275:33297-33301. - PubMed
-
- Bache, R., and N. Pfennig. 1981. Selective isolation of Acetobacterium woodii on methoxylated aromatic acids and determination of growth yields. Arch. Microbiol. 130:255-261.
-
- Beechey, R. B., A. M. Roberton, C. T. Holloway, and I. G. Knight. 1967. The properties of dicyclohexylcarbodiimide as an inhibitor of oxidative phosphorylation. Biochemistry 6:3867-3879. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

