Optical imaging of cancer metastasis to bone marrow: a mouse model of minimal residual disease
- PMID: 11891210
- PMCID: PMC1867183
- DOI: 10.1016/S0002-9440(10)64934-6
Optical imaging of cancer metastasis to bone marrow: a mouse model of minimal residual disease
Abstract
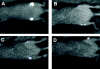
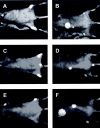
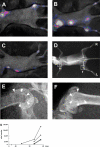
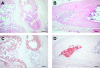
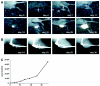
The development of novel anti-cancer strategies requires more sensitive and less invasive methods to detect and monitor in vivo minimal residual disease in cancer models. Bone marrow metastases are indirectly detected by radiography as osteolytic and/or osteosclerotic lesions. Marrow micrometastases elude radiographic detection and, therefore, more sensitive methods are needed for their direct identification. Injection of cancer cells into the left cardiac ventricle of mice closely mimics micrometastatic spread. When luciferase-transfected cells are used, whole-body bioluminescent reporter imaging can detect microscopic bone marrow metastases of approximately 0.5 mm(3) volume, a size below the limit in which tumors need to induce angiogenesis for further growth. This sensitivity translates into early detection of intramedullary tumor growth, preceding the appearance of a radiologically evident osteolysis by approximately 2 weeks. Bioluminescent reporter imaging also enables continuous monitoring in the same animal of growth kinetics for each metastatic site and guides end-point analyses specifically to the bones affected by metastatic growth. This model will accelerate the understanding of the molecular events in metastasis and the evaluation of novel therapies aiming at repressing initial stages of metastatic growth.
Figures

Similar articles
-
Whole-body bioluminescent imaging of human uveal melanoma in a new mouse model of local tumor growth and metastasis.Invest Ophthalmol Vis Sci. 2005 May;46(5):1581-7. doi: 10.1167/iovs.04-0245. Invest Ophthalmol Vis Sci. 2005. PMID: 15851554
-
Bioluminescent human breast cancer cell lines that permit rapid and sensitive in vivo detection of mammary tumors and multiple metastases in immune deficient mice.Breast Cancer Res. 2005;7(4):R444-54. doi: 10.1186/bcr1026. Epub 2005 Apr 8. Breast Cancer Res. 2005. PMID: 15987449 Free PMC article.
-
Severe combined immunodeficient-hu model of human prostate cancer metastasis to human bone.Cancer Res. 1999 Apr 15;59(8):1987-93. Cancer Res. 1999. PMID: 10213511
-
Bone marrow and lymph node assessment for minimal residual disease in patients with breast cancer.Cancer Treat Rev. 2000 Feb;26(1):53-65. doi: 10.1053/ctrv.1999.0150. Cancer Treat Rev. 2000. PMID: 10660491 Review.
-
Bone marrow micrometastases and gastrointestinal cancer detection and significance.Am J Gastroenterol. 2000 Jul;95(7):1644-51. doi: 10.1111/j.1572-0241.2000.02199.x. Am J Gastroenterol. 2000. PMID: 10925963 Review.
Cited by
-
Near-infrared fluorescence molecular imaging of ductal carcinoma in situ with CD44v6-specific antibodies in mice: a preclinical study.Mol Imaging Biol. 2013 Jun;15(3):290-8. doi: 10.1007/s11307-012-0605-8. Mol Imaging Biol. 2013. PMID: 23184608 Free PMC article.
-
A convenient clinically relevant model of human breast cancer bone metastasis.Clin Exp Metastasis. 2008;25(1):33-42. doi: 10.1007/s10585-007-9099-1. Epub 2007 Sep 28. Clin Exp Metastasis. 2008. PMID: 17902030
-
Genetic incorporation of a herpes simplex virus type 1 thymidine kinase and firefly luciferase fusion into the adenovirus protein IX for functional display on the virion.Mol Imaging. 2006 Oct-Dec;5(4):510-9. Mol Imaging. 2006. PMID: 17150163 Free PMC article.
-
Development of New Mouse Breast Cancer Model of Local Bone Metastasis and Verification Using Bisphosphonates.In Vivo. 2022 Mar-Apr;36(2):667-671. doi: 10.21873/invivo.12751. In Vivo. 2022. PMID: 35241520 Free PMC article.
-
Development of a New Focal Mouse Model of Bone Metastasis in Renal Cell Carcinoma.In Vivo. 2024 May-Jun;38(3):1074-1078. doi: 10.21873/invivo.13541. In Vivo. 2024. PMID: 38688604 Free PMC article.
References
-
- Boring CC, Squires TS, Tong T: Cancer statistics, 1993. CA Cancer J Clin 1993, 43:7-26 - PubMed
-
- Jaffe HL: Tumors metastatic to the skeleton. Jaffe HL eds. Tumors and Tumorous Conditions of the Bone and Joints. 1968, :pp 589-618 Lea and Febiger, Philadelphia
-
- Drew M, Dickinson RB: Osseous complications of malignancy. Lokich J eds. Clinical Cancer Medicine, Treatment Tactics. 1980, :pp 97-124 Hall, Boston
-
- Thalmann G, Anezinis P, Devoll R, Farach-Carson C, Chung LWK: Experimental approaches to skeletal metastasis of human prostate cancer. Raghavan D Scher HI Leibel SA Lange PH eds. Principles and Practices of Genitourinary Oncology. 1997, :pp 409-416 Lippincott-Raven, Philadelphia
-
- Willis RA: The spread of tumors in the human body. 1973:pp 229-250 Butterworth, London
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

