Specific entry of Helicobacter pylori into cultured gastric epithelial cells via a zipper-like mechanism
- PMID: 11895977
- PMCID: PMC127843
- DOI: 10.1128/IAI.70.4.2108-2120.2002
Specific entry of Helicobacter pylori into cultured gastric epithelial cells via a zipper-like mechanism
Abstract
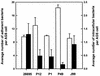
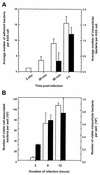
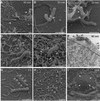
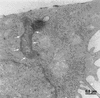
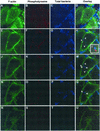
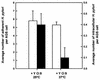
Although Helicobacter pylori has generally been considered an extracellular pathogen, a number of in vitro infection experiments and biopsy examinations have shown that it is capable of occasionally entering mammalian host cells. Here, we characterized this entry process by using AGS cells as a host cell model. In gentamicin protection-invasion assays, the number of H. pylori colonies recovered was lower than that for Salmonella enterica serovar Typhimurium X22, Escherichia coli expressing InvA, and Yersinia enterocolitica YO:9 grown at 25 degrees C but higher than that for Neisseria gonorrhoeae VP1 and Y. enterocolitica YO:9 grown at 37 degrees C. At the ultrastructural level, the entry process was observed to occur via a zipper-like mechanism. Internalized H. pylori was bound in tight LAMP-1-containing vacuoles in close association with condensed filamentous actin and tyrosine phosphorylation signals. Wortmannin, a potent inhibitor of phosphatidylinositol 3-kinase, and calphostin C, an inhibitor of protein kinase C, both inhibited the entry of H. pylori in a sensitive and dose-dependent manner; however, the level of entry was enhanced by sodium vanadate, an inhibitor of tyrosine phosphatases and ATPases. Furthermore, the cytokine tumor necrosis factor alpha antagonized the entry of H. pylori into AGS cells. Collectively, these results demonstrate that the entry of H. pylori into AGS cells occurs via a zipper-like mechanism which involves various host signal transduction events.
Figures


Similar articles
-
Adhesion and Invasion of Gastric Mucosa Epithelial Cells by Helicobacter pylori.Front Cell Infect Microbiol. 2016 Nov 22;6:159. doi: 10.3389/fcimb.2016.00159. eCollection 2016. Front Cell Infect Microbiol. 2016. PMID: 27921009 Free PMC article. Review.
-
Signal transduction-mediated adherence and entry of Helicobacter pylori into cultured cells.Gastroenterology. 1999 Sep;117(3):595-604. doi: 10.1016/s0016-5085(99)70452-x. Gastroenterology. 1999. PMID: 10464135
-
Role of strain type, AGS cells and fetal calf serum in Helicobacter pylori adhesion and invasion assays.FEMS Immunol Med Microbiol. 2000 Sep;29(1):59-67. doi: 10.1111/j.1574-695X.2000.tb01506.x. FEMS Immunol Med Microbiol. 2000. PMID: 10967262
-
Differential activation of mitogen-activated protein kinases in AGS gastric epithelial cells by cag+ and cag- Helicobacter pylori.J Immunol. 1999 Nov 15;163(10):5552-9. J Immunol. 1999. PMID: 10553083
-
Orchestration of aberrant epithelial signaling by Helicobacter pylori CagA.Sci STKE. 2005 Mar 29;2005(277):pe14. doi: 10.1126/stke.2772005pe14. Sci STKE. 2005. PMID: 15798102 Review.
Cited by
-
Establishment of serine protease htrA mutants in Helicobacter pylori is associated with secA mutations.Sci Rep. 2019 Aug 13;9(1):11794. doi: 10.1038/s41598-019-48030-6. Sci Rep. 2019. PMID: 31409845 Free PMC article.
-
Acinetobacter baumannii invades epithelial cells and outer membrane protein A mediates interactions with epithelial cells.BMC Microbiol. 2008 Dec 10;8:216. doi: 10.1186/1471-2180-8-216. BMC Microbiol. 2008. PMID: 19068136 Free PMC article.
-
GSK3β and the control of infectious bacterial diseases.Trends Microbiol. 2014 Apr;22(4):208-17. doi: 10.1016/j.tim.2014.01.009. Epub 2014 Mar 4. Trends Microbiol. 2014. PMID: 24618402 Free PMC article. Review.
-
Type IV secretion of Helicobacter pylori CagA into oral epithelial cells is prevented by the absence of CEACAM receptor expression.Gut Pathog. 2020 May 14;12:25. doi: 10.1186/s13099-020-00363-8. eCollection 2020. Gut Pathog. 2020. PMID: 32435278 Free PMC article.
-
Adhesion and Invasion of Gastric Mucosa Epithelial Cells by Helicobacter pylori.Front Cell Infect Microbiol. 2016 Nov 22;6:159. doi: 10.3389/fcimb.2016.00159. eCollection 2016. Front Cell Infect Microbiol. 2016. PMID: 27921009 Free PMC article. Review.
References
-
- Alm, R. A., and T. J. Trust. 1999. Analysis of the genetic diversity of Helicobacter pylori: the tale of two genomes. J. Mol. Med. 77:834-846. - PubMed
-
- Andersen, L. P., J. Blom, and H. Nielsen. 1993. Survival and ultrastructural changes of Helicobacter pylori after phagocytosis by human polymorphonuclear phagocytes and monocytes. APMIS 101:61-72. - PubMed
-
- Andersson, K., N. Carballeira, K. E. Magnusson, C. Persson, O. Stendahl, H. Wolf-Watz, and M. Fällman. 1996. YopH of Yersinia pseudotuberculosis interrupts early phospho-tyrosine signalling associated with phagocytosis. Mol. Microbiol. 20:1057-1069. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

